SKIN-WHITENING AGENT AND COSMETIC METHOD FOR WHITENING SKIN
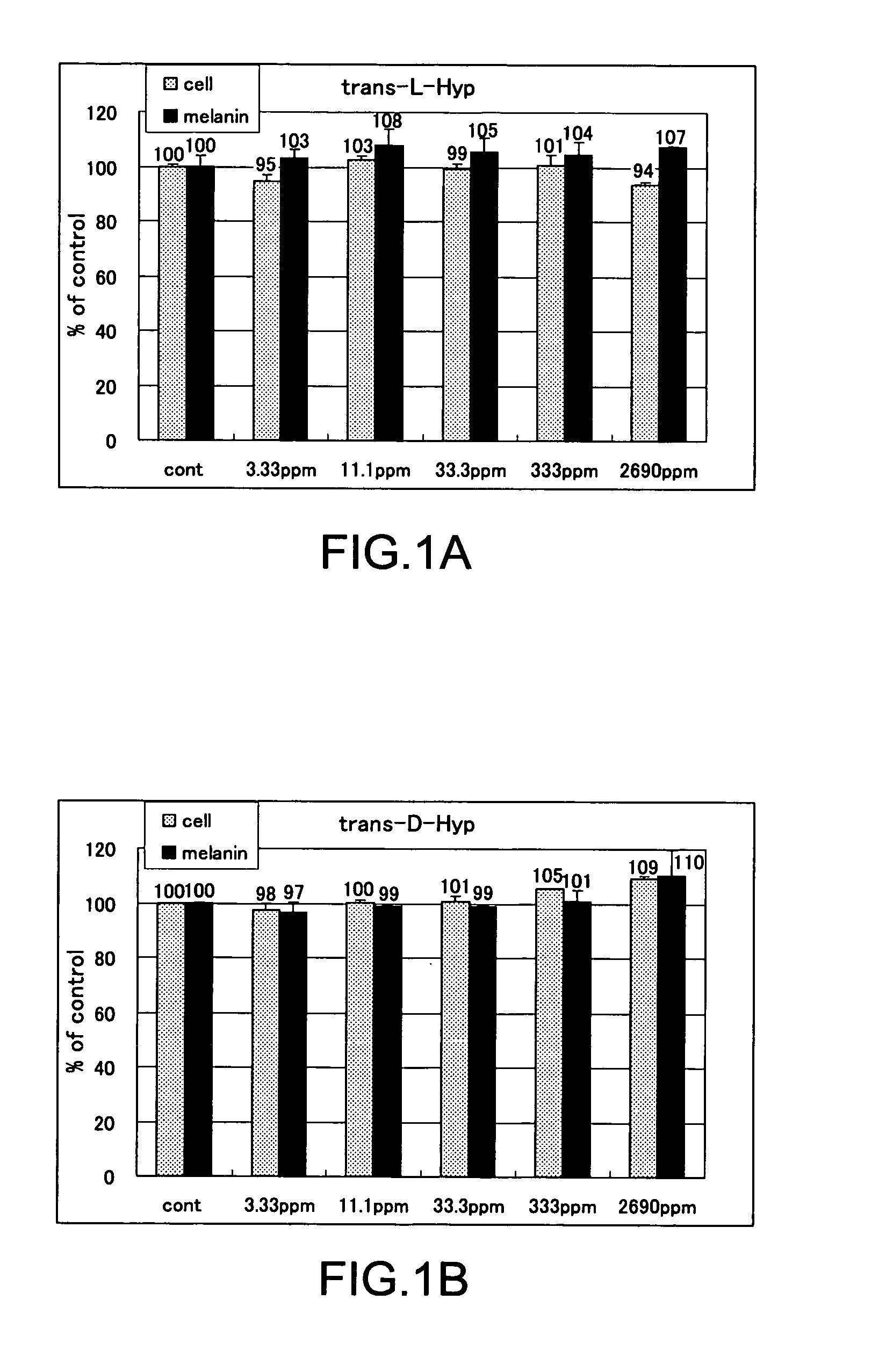
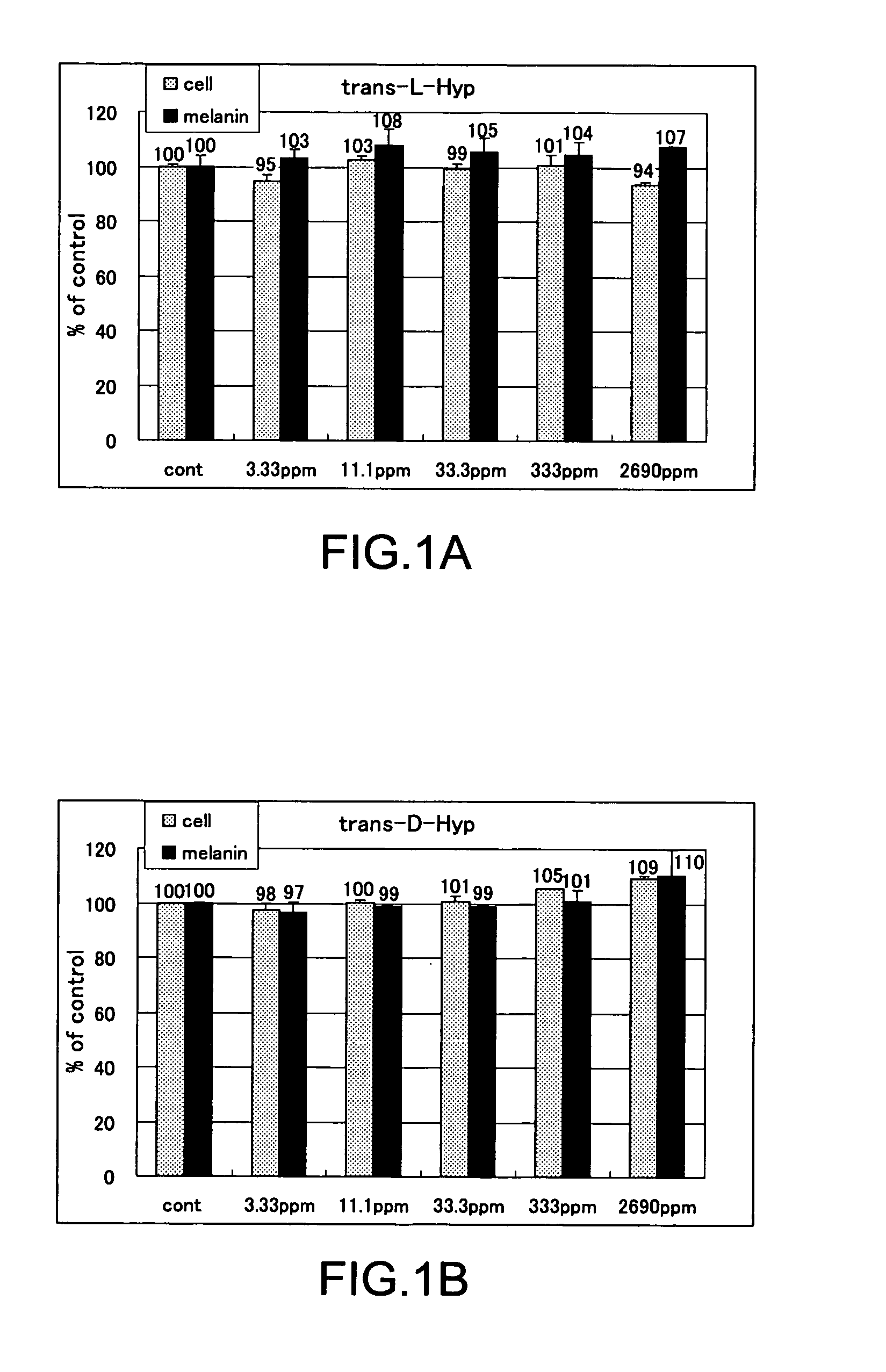
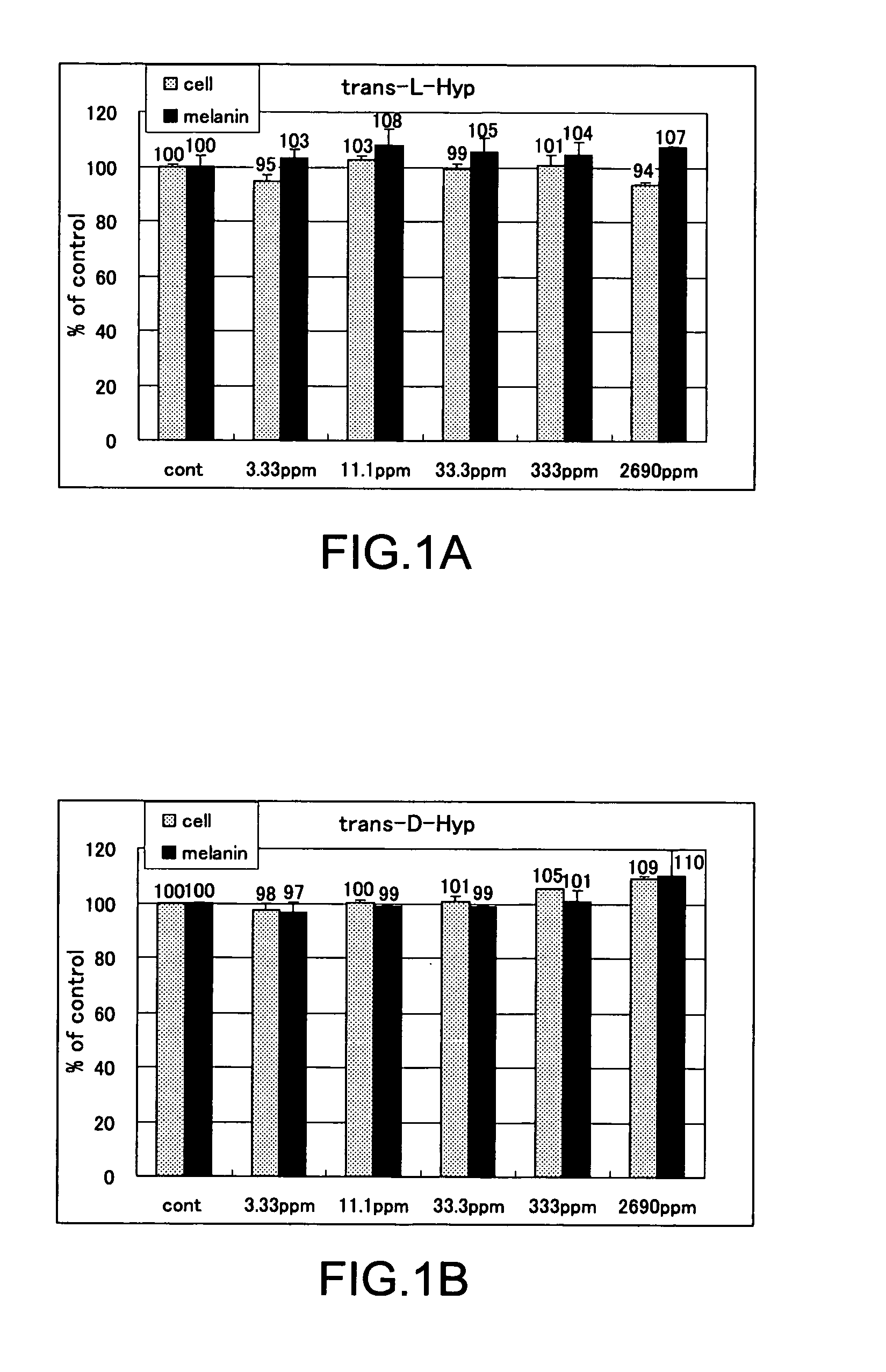
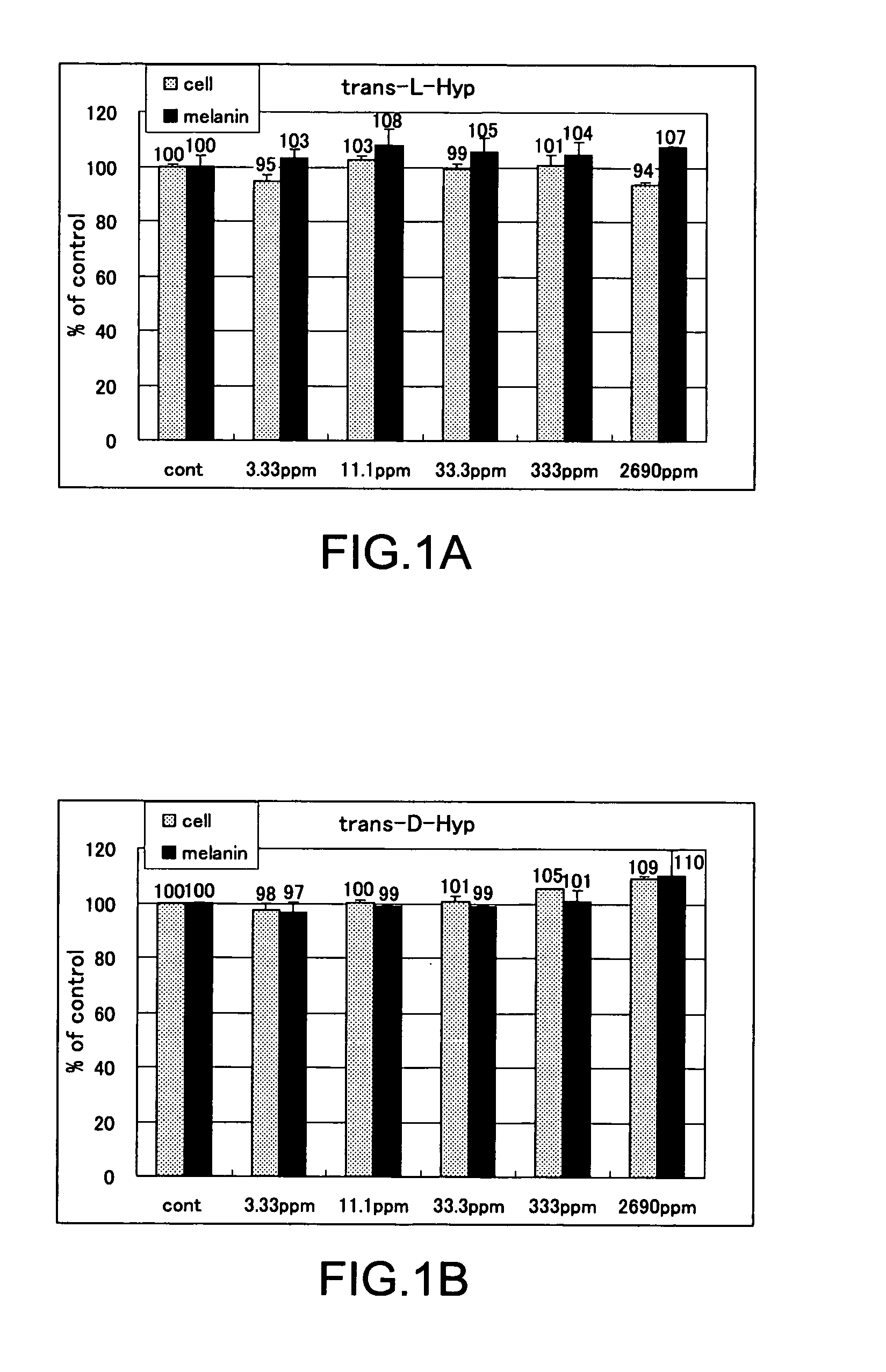
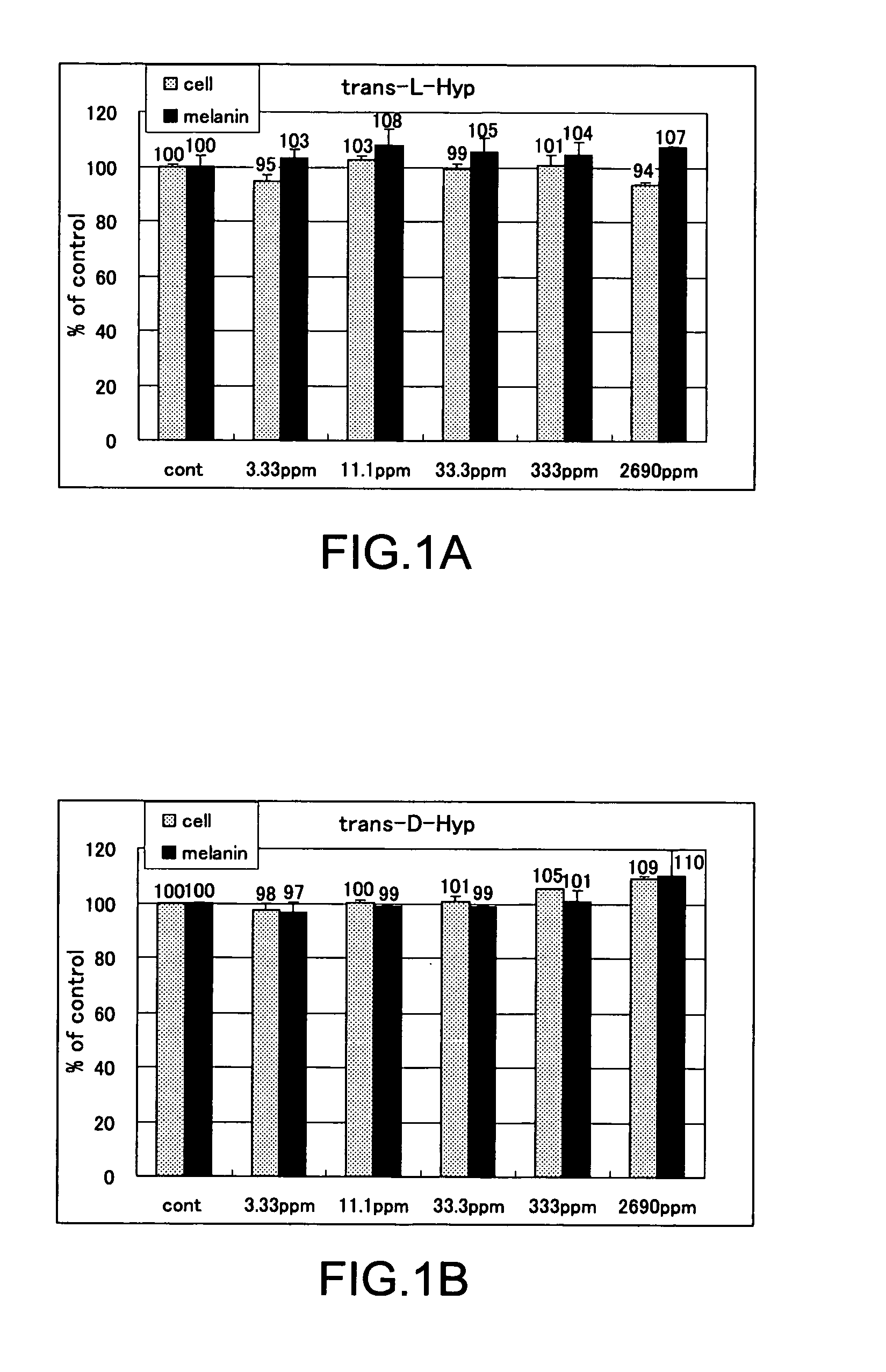
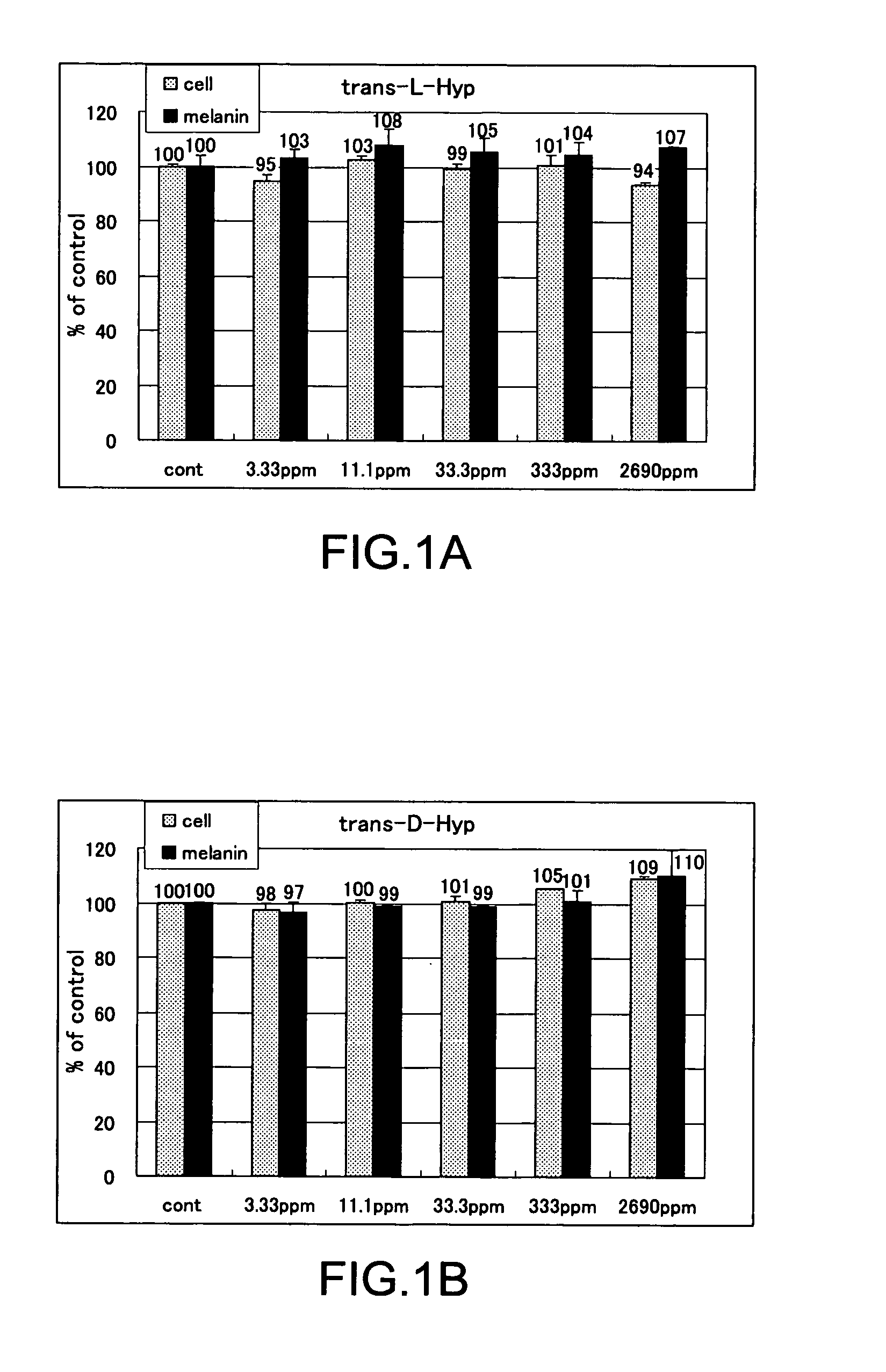
The present invention relates to a skin-whitening agent and a cosmetic method for whitening skin. Specifically, it relates to a skin-whitening agent comprising one or more compounds selected from the group consisting of 4-cis-hydroxyproline and a derivative and/or a salt thereof, and a cosmetic method for whitening the skin comprising a step of administering the skin-whitening agent. Melanin is a skin pigment that is produced by a melanocyte present near the basement membrane of an epidermis. Being surrounded by epidermal cells, it migrates with epidermal cells to skin surface, and eventually discarded as dirt from the skin. Pigment abnormality such as darkening of skin, lentigo, and speckles are caused by abnormal increase or deposition of melanin in the epidermis, and involvement of UV light, female hormones, and genetic causes has been suggested. In this connection, a skin-whitening agent for reducing pigment deposition in skin by inhibiting the production of melanin was developed (Patent Literatures 1 and 2, and Non-Patent Literatures 1 and 2). For example, hydroquinone as a reducing agent has a skin-whitening effect and it is designated as quasi medicines for preventing pigmentation caused by UV light. Further, a compound having an inhibitory activity for tyrosinase, which is an enzyme to catalyze the initial two steps of the reaction for melanin synthesis, is considered as a useful skin-whitening agent. Until now, compounds such as methyl gentisic acid, benzimidazole, and resorcinol have been developed as a skin-whitening agent (Patent Literatures 1 and 2). Hydroquinone is known to have an adverse effect of causing skin irritation, etc. and a mutagenic activity in Chinese hamster V79 cells (Non-Patent Document 1). Although methyl gentisic acid has no such mutagenic property, it has a molecular weight of 168, and therefore there has been a demand to develop a skin-whitening agent which has a lower molecular weight, can be synthesized through a fewer steps and can be produced at low cost. There is also a demand to develop a pharmaceutical product, a cosmetic product, and a food product each of which comprises the skin-whitening agent, and a cosmetic method for whitening the skin including a step of administering the skin-whitening agent. The present invention provides a skin-whitening agent containing one or more compounds selected from the group consisting of 4-cis-hydroxyproline and a derivative and/or a salt thereof. According to the skin-whitening agent of the present invention, the 4-cis-hydroxyproline may be 4-cis-L-hydroxyproline and/or 4-cis-D-hydroxyproline. The present invention provides a cosmetic composition containing the skin-whitening agent of the present invention. The cosmetic composition of the present invention may be a formulation for external application. The present invention provides a food composition containing the skin-whitening agent of the present invention. The present invention provides a pharmaceutical composition containing the skin-whitening agent of the present invention. The pharmaceutical composition of the present invention may be a formulation for external application. The invention provides a cosmetic method for whitening the skin including a step of administering one or more compounds selected from the group consisting of 4-cis-hydroxyproline and a derivative and/or a salt thereof. According to the cosmetic method for whitening the skin, the 4-cis-hydroxyproline may be 4-cis-L-hydroxyproline and/or 4-cis-D-hydroxyproline. According to the cosmetic method for whitening the skin, the step of administering the compound may be external application on skin. According to the cosmetic method for whitening the skin, the step of administering the compound may be oral administration. The invention provides a use of one or more compounds selected from the group consisting of 4-cis-hydroxyproline and a derivative and/or a salt thereof for producing a pharmaceutical composition for treating a disorder accompanied by an abnormality in skin pigmentation. For the application for producing the pharmaceutical composition of the invention, 4-cis-hydroxyproline can be 4-cis-L-hydroxyproline and/or 4-cis-D-hydroxyproline. Hydroxyproline has eight kinds of stereoisomers based on position of hydroxy group, i.e. position 3 or position 4 (position isomer), form of the stereoisomer like cis form or trans form (geometrical isomer), and form of proline like D form or L form (optical isomer). Hydroxyproline is generally present in nature as 4-trans-L-hydroxyproline and very limited presence of other stereoisomers is known. Herein below, structural formula of the geometrical isomers and optical isomers of 4-hydroxyproline is given. [Formula 1] In the detailed description, “hydroxyproline” includes all proline compounds in which position 3 or position 4 of the heterocycle in the L form or D form proline is hydroxylated. Among them, the compounds in which position 4 is hydroxylated include 4-cis-L-hydroxyproline, 4-cis-D-hydroxyproline, 4-trans-L-hydroxyproline and 4-trans-D-hydroxyproline. According to the invention, “4-cis-hydroxyproline” indicates any one of 4-cis-L-hydroxyproline only, 4-cis-D-hydroxyproline only, and 4-cis-L-hydroxyproline and 4-cis-D-hydroxyproline. According to the present invention, “skin-whitening”or “whitens the skin” means that pigment abnormality in skin such as darkening, flecks, and speckles is removed, reduced, and/or prevented. The mechanism of skin whitening includes inhibition of melanocyte differentiation, inhibition of melanin production in melanocyte by inhibiting a melanin producing enzyme such as tyrosinase, degradation, reduction, and depigmentation, etc. of produced melanin, and promoted excretion of melanin in epidermis, but it is not limited thereto. In the detailed description, a “disorder accompanying an abnormality in skin pigmentation” includes, but is not limited, ephelides, chloasma, pigmented nevus+, blue nevus, nevus of Ota, acquired dermal melanocytosis, acropigmentatio reticularis, acromelanosis progressiva, senile spots, chromatosis contact dermatitis, sunburn, pigmentation petaloides actinica, photoluekomelanoderma, chemical-pharmaceutical pigmentation, Addison's disease, pigmentation caused by pregnancy, and Crow-Fukase syndrome, etc. According to the present invention, the derivative of 4-cis-hydroxyproline is defined as that an atom other than the hydrogen atom which constitutes 4-cis-hydroxyproline is covalently bonded to any one of atomic groups under the condition that the skin-whitening effect of 4-cis-hydroxyproline is not reduced. The any one of atomic groups includes, but is not limited to, a protective group such as N-phenyl acetyl group and 4,4′-dimethoxytrityl (DMT) group, biological macromolecules such as protein, peptide, sugar, lipid, and nucleic acid, a synthetic polymer such as polystyrene, polyethylene, polyvinyl, and polyester, and a functional group such as ester group. The ester group may include an aliphatic ester such as methyl ester and ethyl ester or an aromatic ester, for example. According to the present invention, the salt of 4-cis-hydroxyproline means any salt such as metal salt and amine salt, etc. under the condition that the skin-whitening effect of 4-cis-hydroxyproline is not reduced. The metal salt may include an alkaline metal salt and an alkaline earth metal salt. The amine salt may include a triethylamine salt and a benzylamine salt. As shown in the Examples below, 4-cis-hydroxyproline of the present invention as a simple body has, within the concentration range of 33.3 to 333 ppm or 11.1 to 33.3 ppm, an effect of reducing melanin content in cultured mouse B16 melanoma cells. As such, the amount of 4-cis-hydroxyproline contained in the pharmaceutical composition, cosmetic composition, and food composition of the invention may be any content as long as 4-cis-hydroxyproline itself within the concentration range above is delivered to a melanocyte in a skin tissue of a living organism. Where the composition of the invention is prepared as an external application, the content of 4-cis-hydroxyproline may be from 0.000015% by weight to 50% by weight compared to the total weight of the composition of the invention, or it may be within the range having an upper value of weight concentration at which the compound may be contained in maximum amount. Specifically, when the composition of the invention is an external formulation, the content of 4-cis-hydroxyproline is preferably 0.00003% by weight to 30% by weight, and most preferably 0.0003% by weight to 3% by weight. When the composition of the invention is a formulation for internal application, the content of 4-cis-hydroxyproline may be within the range of 0.00001% by weight to 100% by weight. When the composition of the invention is a formulation for internal application, the content of 4-cis-hydroxyproline is preferably 0.00002% by weight to 80% by weight, and most preferably, 0.0002% by weight to 60% by weight. The lower intake limit per day of 4-cis-hydroxyproline in the composition of the invention may be 0.01 ng, 0.1 ng, or 1 ng per kg of body weight. The cosmetic composition of the invention may be formulated appropriately as required with other components that are used for a cosmetic product such as quasi drugs or an external formulation for skin such as pharmaceuticals under the condition that the skin-whitening effect of 4-cis-hydroxyproline is not reduced. Examples of the other components (i.e. optionally added components) include an oil, a detergent, powder, a colorant, water, alcohols, a viscosity improver, a chelating agent, silicone, an anti-oxidant, a UV absorbing agent, a moisturizing agent, a flavouring agent, variety of pharmaceutical compounds, a preservative, a pH adjustment agent, and a neutralizing agent or the such as. The cosmetic composition of the invention may be any one of conventionally used external formulations for skin and those used as a cosmetic composition such as ointment, cream, emulsion, lotion, pack, and bathing product, and the formulation type is not specifically limited. The food composition of the invention may include, in addition to 4-cis-hydroxyproline itself, a salt of 4-cis-hydroxyproline, and/or its derivative which can release 4-cis-hydroxyproline by an enzyme for metabolizing a drug in a living body, a component which is allowed for a food product such as a flavoring agent, a coloring agent, and a preservative under the condition that the skin-whitening effect of 4-cis-hydroxyproline is not inhibited. Examples of the food composition of the invention include, but is not limited to, any of those conventionally used as a food composition such as beverages, gummy candy, candy, and biscuit. The pharmaceutical composition of the invention may contain, in addition to 4-cis-hydroxyproline itself, a salt of 4-cis-hydroxyproline, and/or its derivative which can release 4-cis-hydroxyproline by an enzyme for metabolizing a drug in a living body, one or more of other components which are effective for treating a disorder and/or pharmaceutically acceptable additives under the condition that the skin-whitening effect of 4-cis-hydroxyproline is not inhibited. The additives include a diluting agent, a swelling agent, a binding agent, adhesives, a lubricating agent, a fluidity promoting agent, a plasticizing agent, a disintegrating agent, a carrier solvent, a buffer agent, a coloring material, a flavor, a sweetening agent, a preservative, a stabilizing agent, an adsorbing agent, and other additives for pharmaceuticals that are known to those skilled in the art, but they are not limited thereto. The Examples given below are only to illustrate the invention, and the scope of the invention is not limited by them. The scope of the invention is defined only by the working of the claims. Mouse B16 melanoma cell was used. The cells were inoculated at 1×105cells/well to a 6-well plate, and cultured with using a commercially available medium (trade name: EAGLE MEM, manufactured by Nissui Pharmaceutical Co., Ltd.) supplemented with 10% fetal bovine serum. The cells were cultured at 37° C., 5% CO2and under completely saturated water-vapor atmosphere. Reagents Among the hydroxyprolines used for the Examples of the invention, 4-cis-L-hydroxyproline, 4-cis-D-hydroxyproline and 4-trans-L-hydroxyproline were purchased from Sigma-Aldrich Japan, and 4-trans-D-hydroxyproline was purchased from Bachem. Addition of Hydroxyproline For determining the effect of hydroxyproline on cell growth and melanin content reduction, after culturing the cells for 24 hours, stock solutions of 4-trans-L-hydroxyproline, 4-trans-D-hydroxyproline, 4-cis-L-hydroxyproline or 4-cis-D-hydroxyproline were prepared and added in the same volume to the cells, further cultured for 3 days and used for the experiment. Meanwhile, as a control group, the same volume of water was added. Quantification of the Number of Cells 3 days after the addition of a solution of test compound, the medium was removed by suction. Eagle's-MEM containing the solution of 10% alamarBlue (trade name: BIOSOURCE, manufactured by Biosource International) was added and the reaction was allowed to occur at 37° C. Thirty minutes later, the fluorescence was measured with excitation wavelength of 544 nm and measurement wavelength of 590 nm. By having the obtained number as a relative value of the cell number, the ratio of number of cells (i.e. % number of cells) for a group added with the test compound compared to a group not added with the test compound (i.e. only the solvent was added) was calculated. Higher % number of cells indicates lower cell toxicity. When it is less than 80%, it was determined as “toxic.” Results of Quantifying the Number of Cells In Quantification of Melanin The medium was removed by suction. After washing three times with the buffer (phosphate buffer solution 50 mM, pH 6.8), the cells were lysed by addition of 1 N NaOH, and the absorbance was measured at 475 nm. By having the obtained value as a relative value of the melanin content, the ratio of melanin content (%) for a group added with the test compound compared to a group not added with the test compound (i.e. only the solvent was added) was calculated. Lower ratio of melanin content means the higher inhibition effect on melanin production. Determination of Melanin Content By using the measured value of melanin content that was obtained according to the measurement method above, percentage ratio of the melanin content was calculated by dividing the melanin content in the cells added with 4-trans-L-hydroxyproline, 4-trans-D-hydroxyproline, 4-cis-L-hydroxyproline, or 4-cis-D-hydroxyproline by the melanin content in the cells for control experiment, and the resulting value was taken as a ratio of melanin content. Results of Melanin Quantification In Experiments for Quantifying Cell Growth and Melanin Content by Using a Mixture Solution of 4-Trans-L-Hydroxyproline and 4-Cis-D-Hydroxyproline The equal amounts of 4-trans-L-hydroxyproline and 4-cis-D-hydroxyproline were mixed to produce the mixture solution of 4-trans-L-hydroxyproline and 4-cis-D-hydroxyproline, which was then provided for the experiments. Cell culture and quantification and evaluation of number of the cells and melanin were carried out in the same manner as the Examples above. Results of Cell Growth Test Using Mixture Solution In Results of Quantifying Melanin Using the Mixture Solution In Test for Determining Cell Growth and Melanin Content Reduction Using a Mixture at Various Mixing Ratio By using the mixture solution in which 4-trans-L-hydroxyproline and 4-cis-D-hydroxyproline were mixed at various ratio, test for determining cell growth and melanin content reduction was carried out. Cell culture and quantification and evaluation were carried out in the same manner as the Examples above. Results of Cell Growth Test Using the Mixture Solution at Various Mixing Ratio In Results of Melanin Quantification Using the Mixture Solution at Various Mixing Ratio In Mix examples including an emulsion formulation, a skin patch, a tablet, a soft capsule, a granule, a drink, a candy, a cookie, soybean paste, a French dressing, mayonnaise, baguette, soy sauce, furikake (a seasoned powder for sprinkling over rice), seasoning and natto (Japanese fermented soybean paste) sauce, natto, non-filtered dark vinegar, a cream, a body cream, a gel formulation, a peel-off mask, an impregnation mask, an emulsion, a cosmetic water, and an aerosol, all containing 4-cis-hydroxyproline according to the invention, are given below. However, the mix examples are listed only for illustration and it is not intended that the scope of the invention is limited by them. Method for Preparation of Mix Example 10 (Cookie) Granulated sugar is gradually added to butter under mixing. Egg, decapeptide, GABA, and the flavor are further added and mixed. After mixing thoroughly, soft flour obtained by uniformly sieving is added to the mixture, which is then mixed at low speed. The agglomerated mixture is kept in a refrigerator. After molding, cookies are obtained by baking for 15 minutes at 170° C. Method for Preparation of Mix Example 11 (Soybean Paste) Rice yeast and salt are mixed well with each other. Washed soybeans are soaked in water (×3 volume) overnight. Water is removed, and the soybeans are cooked while adding fresh water and then placed in a basket. The cooking water (i.e. filtered water) is collected and 4-cis-hydroxyproline is dissolved therein to 10% w/v. The cooked soybeans are immediately smashed and added with the rice yeast mixed with salt. While adding thereto the filtered water in which 4-cis-hydroxyproline is dissolved, the beans are thoroughly mixed until clay-such as hardness is obtained. The mixture is rolled to have a meat ball shape, and the resultant is filled in a container from end to end. After making the surface flat, the container is sealed by covering with a plastic wrap. The container is changed three months later, and again, after making the surface flat, the container is sealed by covering with a plastic wrap. Meanwhile, in addition to adding 4-cis-hydroxyproline to filtered water, rice yeast which can produce a large amount of 4-cis-hydroxyproline can also be used. In addition, 4-cis-hydroxyproline or its salt can be added to commercially available soybean paste. Method for Preparation of Mix Example 12 (French Dressing) To vinegar, sodium chloride and 4-cis-hydroxyproline are added and stirred well for dissolution. Salad oil is added thereto and pepper is added under thorough mixing. Method for Preparation of Mix Example 13 (Mayonnaise) To egg yolk (room temperature), vinegar, sodium chloride, 4-cis-hydroxyproline, and pepper are added and the mixed sufficiently by using a whipping machine. The mixing is further continued while adding salad oil in small portions to obtain an emulsion. Finally, sugar is added and mixed. Method for Preparation of Mix Example 14 (Baguette) To warm water, 1 g of sugar and dry yeast are added for pre-fermentation. Strong flour, soft flour, sodium chloride, 5 g of sugar, and 4-cis-hydroxyproline are added to a bowl, and the pre-fermented yeast is added thereto. After sufficient kneading, the mixture is formed into a ball, and subjected to primary fermentation at 30° C. The dough is kneaded again, kept for a while, and then formed into an appropriate shape. Final fermentation is carried out by using an electronic fermentation machine. After forming coupes, baking is carried out in an oven at 220° C. for 30 minutes. Method for Preparation of Mix Example 15 (Soy Sauce) To commercially available soy sauce, 4-cis-hydroxyproline is added and stirred well. Meanwhile, in addition to adding 4-cis-hydroxyproline or its salt, soy sauce can be produced by using yeast which can produce a large amount of 4-cis-hydroxyproline. Method for Preparation of Mix Example 16 (Yogurt) Fermentation is carried out at 40° C. to 45° C. Other commercially available starter cultures can be used and also 4-cis-hydroxyproline can be added to commercially available yogurt. In addition, instead of adding 4-cis-hydroxyproline or its salt, a microorganism which can produce a large amount of 4-cis-hydroxyproline can be used. Method for Preparation of Mix Example 19 (Natto) Instead of adding 4-cis-hydroxyproline or its salt, a microorganism which can produce a large amount of 4-cis-D-hydroxyproline can be used for producing natto. Method for Preparation of Mix Example 20 (Non-Filtered Dark Vinegar) Instead of adding 4-cis-hydroxyproline or its salt, a microorganism which can produce a large amount of 4-cis-D-hydroxyproline can be used for producing vinegar, dark vinegar, and non-filtered dark vinegar. Method of Filling Mix Example 31 (Aerosol Urea Spraying Agent) An aerosol urea stock solution for external application and dimethyl ether are filled in a pressure-resistant aerosol aluminum container having an inner surface coated with Teflon (registered trademark) to produce an aerosol formulation. The development of a skin-whitening agent which has a lower molecular weight, can be synthesized through a fewer steps and can be produced at low cost has been demanded. Thus, it is provided by the present invention a skin-whitening agent including one or more compounds selected from the group consisting of 4-cis-hydroxyproline and its derivative and/or its salt, a skin-whitening composition for external formulation to the skin, a cosmetic composition and a pharmaceutical composition, each of which contains the skin-whitening agent of the invention, and a cosmetic method for whitening the skin, which includes a step of administering one or more compounds selected from the group consisting of 4-cis-hydroxyproline and its derivative and/or its salt. In the skin-whitening agent and the cosmetic method for whitening the skin of the invention, the 4-cis-hydroxyproline may be 4-cis-L-hydroxyproline and/or 4-cis-D-hydroxyproline. 1. A skin-whitening agent comprising one or more compounds selected from the group consisting of 4-cis-hydroxyproline and a derivative and/or a salt thereof. 2. The skin-whitening agent according to 3. The skin-whitening agent according to 4. The skin-whitening agent according to 5. A skin-whitening composition for external formulation to skin comprising the skin-whitening agent according to 6. A food composition comprising the skin-whitening agent according to 7. A pharmaceutical composition comprising the skin-whitening agent according to 8. A composition for external formulation to skin comprising one or more compounds selected from the group consisting of 4-cis-hydroxyproline and a derivative and/or a salt thereof. 9. A cosmetic method for whitening skin comprising a step of administering one or more compounds selected from the group consisting of 4-cis-hydroxyproline and a derivative and/or a salt thereof. 10. The method according to 11. The method according to TECHNICAL FIELD
BACKGROUND ART
PRIOR ART DOCUMENTS
Non-Patent Documents
DISCLOSURE OF THE INVENTION
Problems to be Solved by the Invention
Means for Solving the Problems
BRIEF DESCRIPTION OF THE DRAWINGS
DESCRIPTION OF EMBODIMENTS
Example 1
Cell Culture
Mix Example 1
Emulsion Formulation
4-cis-hydroxyproline 0.42 behenyl alcohol 0.2 cetanol 0.5 glycerin monofatty acid ester 1.8 hydrogenated castor oil POE (60) 1.0 white vaseline 2.0 fluid paraffin 10.0 isopropyl myristate 3.0 methyl polysiloxane (6 cs) 1.5 conc. glycerin 13.0 dipropylene glycol 2.0 carboxyvinyl polymer 0.25 sodium hyaluronic acid 0.005 potassium hydroxide q.s. lactic acid q.s. sodium edetate q.s. ethyl paraben q.s. purified water remainder 100.000 Mix Example 2
Skin Patch
4-cis-hydroxyproline 0.3 polyacrylic acid 3.0 sodium polyacrylate 2.5 gelatin 0.5 sodium carboxymethyl cellulose 4.0 polyvinyl alcohol 0.3 conc. glycerin 14.0 1,3-butylene glycol 12.0 aluminum hydroxide 0.1 sodium edetate 0.03 methyl paraben 0.1 purified water remainder 100.00 Mix Example 3
Tablet
4-cis-hydroxyproline 360.5 lactose 102.4 calcium carboxymethyl 29.9 cellulose hydroxypropyl cellulose 6.8 magnesium stearate 5.2 crystalline cellulose 10.2 515.0 Mix Example 4
Tablet
sucrose ester 70 crystalline cellulose 74 methyl cellulose 36 glycerin 25 4-cis-hydroxyproline 475 N-acetyl glucosamine 200 hyaluronic acid 150 vitamin E 30 vitamin B6 20 vitamin B2 10 α-lipoic acid 20 coenzyme Q10 40 ceramide (devil's tongue jelly 50 extract) L-proline 300 1500 Mix Example 5
Soft Capsule
soybean oil for cooking 530 50 50 4-cis-hydroxyproline 100 royal jelly 50 maca 30 GABA 30 beeswax 60 gelatin 375 glycerin 120 glycerin fatty acid ester 105 1500 Mix Example 6
Soft Capsule
brown rice germ oil 659 4-cis-hydroxyproline 500 resveratrol 1 lotus germ extract 100 elastin 180 DNA 30 folic acid 30 1500 Mix Example 7
Granule
4-cis-hydroxyproline 400 vitamin C 100 soybean isoflavone 250 reduced lactose 300 soybean oligosugar 36 erythritol 36 dextrin 30 flavor 24 citric acid 24 1200 Mix Example 8
Drink
1.6 1.6 4-cis-hydroxyproline 1.6 reduced maltose syrup 28 erythritol 8 citric acid 2 flavor 1.3 N-acetyl glucosamine 1 hyaluronic acid Na 0.5 vitamin E 0.3 vitamin B6 0.2 vitamin B2 0.1 α-lipoic acid 0.2 coenzyme Q10 1.2 ceramide (devil's tongue 0.4 jelly extract) L-proline 2 purified water remainder 60 Mix Example 9
Candy
sugar 50 syrup 48 4-cis-hydroxyproline 1 flavor 1 100 Mix Example 10
Cookie
soft flour 45.0 butter 17.5 granulated sugar 20.0 4-cis-hydroxyproline 4.0 egg 12.5 flavor 1.0 100.0 Mix Example 11
Soybean Paste
soybean 1000 rice yeast 1000 salt 420 4-cis-hydroxyproline 158 water remainder 4000 Mix Example 12
French Dressing
salad oil 27.0 vinegar 30.0 sodium chloride 0.9 4-cis-hydroxyproline 1.1 pepper 1.0 60.0 Mix Example 13
Mayonnaise
salad oil 134.0 vinegar 5 sodium chloride 0.9 4-cis-hydroxyproline 1 egg yolk 18 sugar 0.2 pepper 0.9 160.0 Mix Example 14
Baguette
strong flour 140 soft flour 60 sodium chloride 3 sugar 6 4-cis-hydroxyproline 2 dry yeast 4 warm water 128 343 Mix Example 15
Soy Sauce
commercially available soy 990 sauce 4-cis-hydroxyproline 10 1000 Mix Example 16
Yogurt
milk 880 50 50 4-cis-hydroxyproline 20 1000 Mix Example 17
Furikake
4-cis-hydroxyproline 50 seaweed 15 sodium L-glutamate 10 sodium chloride 2 roasted sesame seed 10 thinly sliced dried mackerel 10 sugar 1 soy sauce 2 100 Mix Example 18
Seasoning and Natto Sauce
commercially available 9.9 natto sauce 4-cis-hydroxyproline 0.1 10 Mix Example 19
Natto
commercially available natto 19.9 4-cis-hydroxyproline 0.1 20 Mix Example 20
Non-Filtered Dark Vinegar
commercially available 950 non-filtered dark vinegar 4-cis-hydroxyproline 50 1000 Mix Example 21
Cream
fluid paraffin 3 vaseline 1 dimethyl polysiloxane 1 stearyl alcohol 1.8 behenyl alcohol 1.6 glycerin 8 dipropylene glycol 5 2 hydrogenated oil 3 squalene 6 stearic acid 2 cholesteryl hydroxystearate 0.5 cetyl 2-ethylhexanoate 4 polyoxyethylene hydrogenated 0.5 castor oil self-emulsifying glycerin 3 monostearate potassium hydroxide 0.15 sodium hexametaphosphate 0.05 trimethyl glycine 2 α-tocopherol 2-L-ascorbic acid phosphoric acid diester 1 potassium tocopherol acetate 0.1 4-cis-hydroxyproline 4 paraben q.s. trisodium edetate 0.05 4-t-butyl-4′-methoxy 0.05 dibenzoylmethane diparamethoxy cinnamic acid mono-2-ethylhexanoic acid 0.05 glyceryl coloring agent q.s. carboxyvinyl polymer 0.05 purified water remainder 100.00 Mix Example 22
Body Cream
dimethyl polysiloxane 3 decamethyl cyclopentasiloxane 13 dodecamethyl cyclohexasiloxane 12 polyoxyethylene-methyl 1 polysiloxane copolymer ethanol 2 isopropanol 1 glycerin 3 dipropylene glycol 5 polyethylene glycol 6000 5 sodium hexametaphosphate 0.05 tocopherol acetate 0.1 4-cis-hydroxyproline 5 fennel extract 0.1 0.1 0.1 L-menthol q.s. paraoxybenzoic acid ester q.s. trisodium edetate 0.05 dimorpholino pyridazinone 0.01 methylbis(trimethylsiolxy)silyl 0.1 isopentyl trimethoxycinnamate iron sulfate q.s. cobalt titanate q.s. dimethyl distearyl ammonium 1.5 hectorite polyvinyl alcohol 0.1 hydroxyethyl cellulose 0.1 trimethylsiloxy silicate 2 flavor q.s. purified water remainder 100.00 Mix Example 23
Gel Formulation
dimethyl polysiloxane 5 glycerin 2 1,3-butylene glycol 5 polyethylene glycol 1500 3 polyethylene glycol 20000 3 cetyl octanoate 3 citric acid 0.01 sodium citrate 0.1 sodium hexametaphosphate 0.1 dipotassium glycyrrhiziate 0.1 4-cis-hydroxyproline 2 tocopherol acetate 0.1 skullcap extract 0.1 strawberry geranium extract 0.1 trisodium edetate 0.1 xanthan gum 0.3 acrylate-alkyl methacrylate 0.05 copolymer (PEMULEN TR-2) agar powder 1.5 phenoxyethanol q.s. dibutylhydroxy toluene q.s. purified water remainder 100.00 Mix Example 24
Peel-Off Mask
ethanol 10 1,3-butylene glycol 6 polyethylene glycol 4000 2 olive oil 1 1 phytosteryl hydroxystearate 0.05 lactic acid 0.05 sodium lactate 0.1 L-ascorbic acid disodium sulfuric 0.1 acid ester α-tocopherol 2-L-ascorbic acid 0.1 phosphoric acid diester potassium 4-cis-hydroxyproline 10 fish collagen 0.1 chondroitin sodium sulfate 0.1 sodium carboxymethyl cellulose 0.2 polyvinyl alcohol 12 paraoxy benzoic acid ester q.s. flavor q.s. purified water remainder 100.00 Mix Example 25
Impregnation Mask
glycerin 1 1,3-butylene glycol 8 xylit 2 polyethylene glycol 1500 2 rosemary oil 0.01 sage oil 0.1 citric acid 0.02 sodium citrate 0.08 sodium hexametaphosphate 0.01 hydroxypropyl-β-cyclodextrin 0.1 4-cis-hydroxyproline 0.5 birch extract 0.1 lavender oil 0.01 xanthan gum 0.05 carboxyvinyl polymer 0.15 paraoxy benzoic acid ester q.s. purified water remainder 100.00 Mix Example 26
Emulsion
fluid paraffin 7 vaseline 3 decamethyl cyclopentasiloxane 2 behenyl alcohol 1.5 glycerin 5 dipropylene glycol 7 polyethylene glycol 1500 2 jojoba oil 1 isostearic acid 0.5 stearic acid 0.5 behenic acid 0.5 tetra 2-ethylhexanoic acid 3 pentaerythritol cetyl 2-ethylhexanoate 3 monostearic acid glycerin 1 monostearic acid polyoxyethylene 1 glycerin potassium hydroxide 0.1 sodium hexametaphosphate 0.05 stearyl glycyrrhetate 0.05 4-cis-hydroxyproline 1 royal jelly extract 0.1 yeast extract 0.1 tocopherol acetate 0.1 sodium acetylated hyaluronic acid 0.1 trisodium edetate 0.05 4-t-butyl-4′-methoxy dibenzoylmethane 0.1 2-ethylhexyl paramethoxycinnamate 0.1 carboxyvinyl polymer 0.15 paraben q.s. flavor q.s. purified water remainder 100.00 Mix Example 27
Emulsion
dimethyl polysiloxane 2 behenyl alcohol 1 batyl alcohol 0.5 glycerin 5 1,3-butylene glycol 7 erythritol 2 hydrogenated oil 3 squalene 6 tetra-2-ethylhexanoic acid 2 pentaerythritol isostearic acid 1 polyoxyethylene glyceryl monostearic acid 1 polyoxyethylene glycerin 4-cis-hydroxyproline 0.3 potassium hydroxide q.s. sodium hexametaphosphate 0.05 phenoxyethanol q.s. carboxyvinyl polymer 0.1 purified water remainder 100.00 Mix Example 28
Cosmetic Water
ethyl alcohol 5 glycerin 1 1,3-butylene glycol 5 polyoxyethylene polyoxypropylene decyl 0.2 tetradecyl ether sodium hexametaphosphate 0.03 trimethyl glycine 1 sodium polyasparaginic acid 0.1 α-tocopherol 2-L-ascorbic acid phosphoric acid diester 0.1 potassium thiotaurine 0.1 4-cis-hydroxyproline 8 trisodium EDTA 0.1 carboxyvinyl polymer 0.05 potassium hydroxide 0.02 phenoxyethanol q.s. flavor q.s. purified water remainder 100.00 Mix Example 29
Cosmetic Water
ethanol 10 dipropylene glycol 1 polyethylene glycol 1000 1 polyoxyethylene 1 methylglucoside jojoba oil 0.01 tri-2-ethylhexanoic acid 0.1 glyceryl polyoxyethylene hydrogenated 0.2 castor oil diisostearic acid polyglyceryl 0.15 sodium N-stearoyl-L-glutamate 0.1 citric acid 0.05 sodium citrate 0.2 potassium hydroxide 0.4 dipotassium glycyrrhiziate 0.1 arginine hydrochloride 0.1 L-ascorbic acid 2-glucoside 2 4-cis-hydroxyproline 0.5 trisodium edetate 0.05 2-ethylhexyl paramethoxy 0.01 cinnamate dibutylhydroxy toluene q.s. paraben q.s. deep sea water 3 flavor q.s. purified water remainder 100.00 Mix Example 30
Urea Aerosol Stock Solution for External Application
ethanol 15.0 polyoxyethylene hydrogenated 1.5 castor oil 50 diphenhydramine 1.0 dibucaine 2.0 tocopherol acetate 0.5 4-cis-hydroxyproline 0.1 isostearic acid 0.1 1,3-butylene glycol 3.0 polyethylene glycol 400 3.0 camphor 0.05 urea 20.0 purified water remainder 100.00 Mix Example 31
Aerosol Urea Spraying Agent
urea aerosol stock 65.0 solution for external application dimethyl ether 35.0 100.00