METHODS FOR PRODUCTION OF OXYGENATED TERPENES
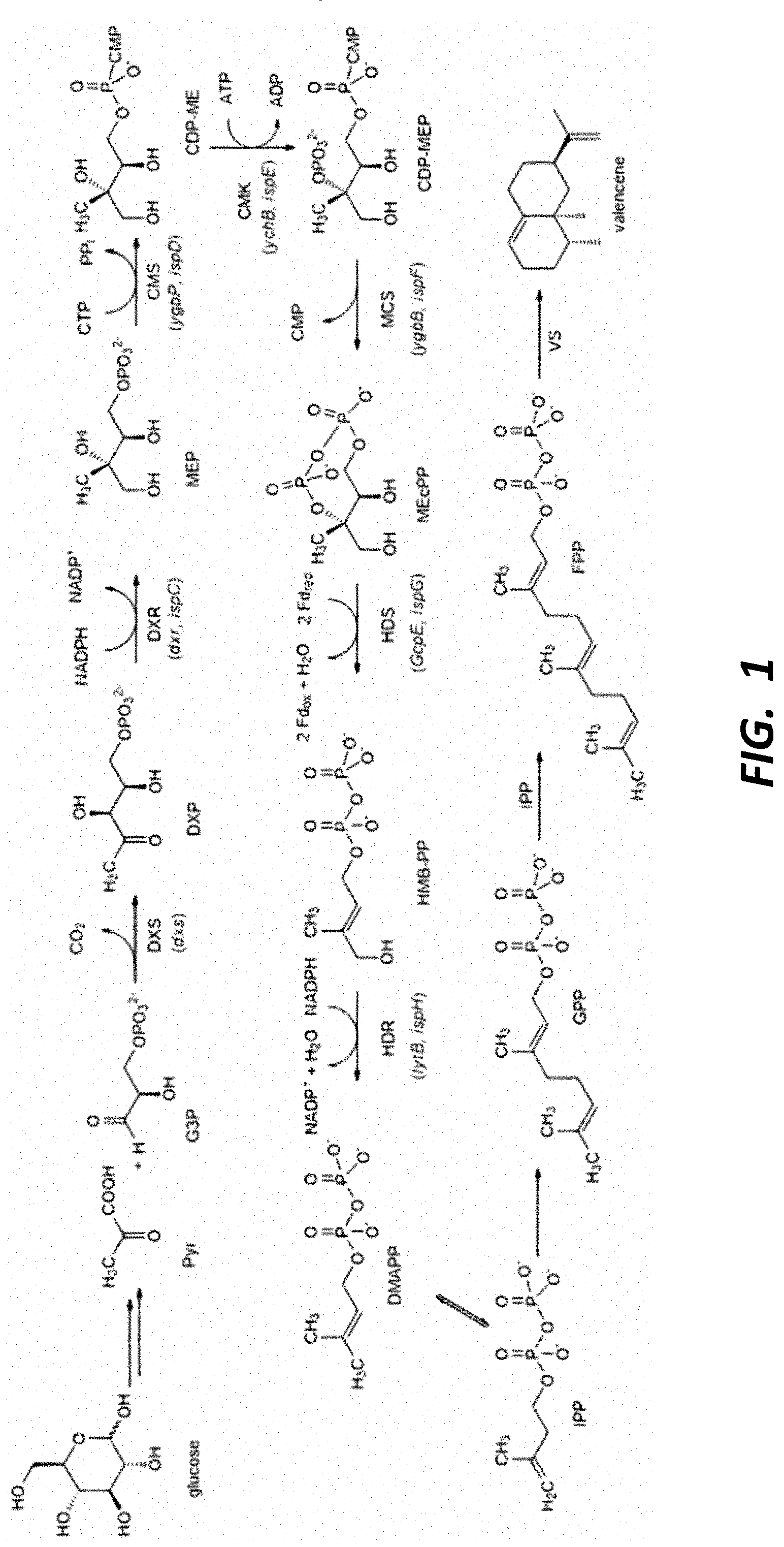
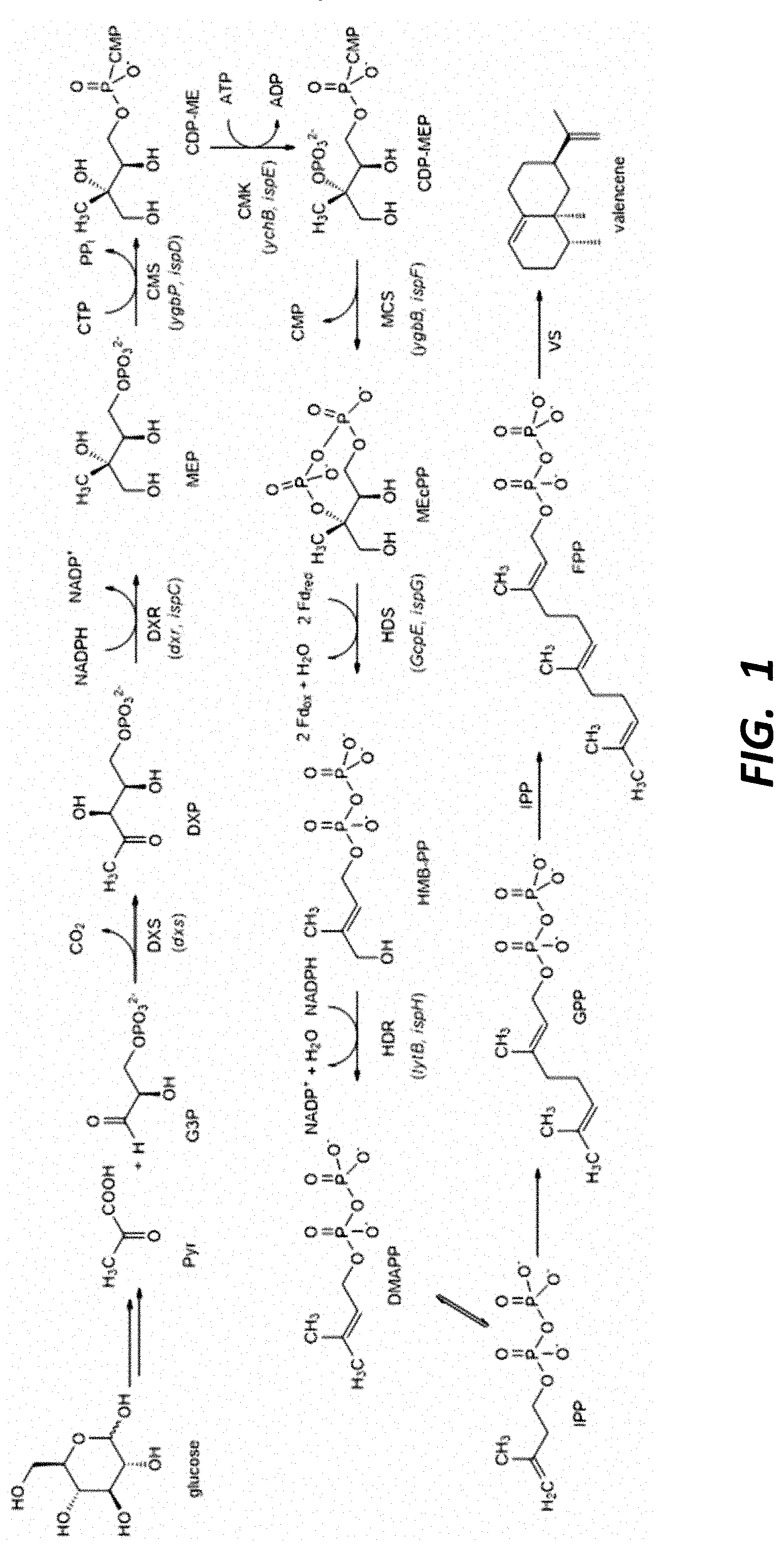
This application is a continuation of U.S. patent application Ser. No. 16/669,051, filed on Oct. 30, 2019, which is a divisional of U.S. patent application Ser. No. 15/505,022, filed Feb. 17, 2017, which is a United States National Stage Application of International Application No. PCT/US2015/046421, filed Aug. 21, 2015, which claims the benefit of, and priority to, U.S. Provisional Application No. 62/040,284, filed Aug. 21, 2014, each of which is incorporated herein by reference in its entirety. The present disclosure relates to oxygenated sesquiterpenes (e.g., nootkatone and/or nootkatol) and methods for their production and use. The disclosure also provides enzymes for the production of oxygenated sesquiterpenes (e.g., nootkatone and/or nootkatol) and methods for identifying, selecting, making and using these enzymes. The food and beverage industries as well as other industries such as the perfume, cosmetic and health care industries routinely use terpenes and/or terpenoid products as flavours and fragrances. By way of example, many sesquiterpene compounds are used in perfumery (e.g., patchoulol) and in the flavour industry (e.g., nootkatone) and many are extracted from plants. However, factors such as: (i) the availability and high price of the plant raw material; (ii) the relatively low terpene content in plant; and (iii) the tedious and inefficient extraction processes to produce sufficient quantities of terpene products on an industrial scale all have stimulated research on the biosynthesis of terpenes using plant-independent systems. Consequently, effort has been expended in developing technologies to engineer microorganisms for converting renewable resources such as glucose into terpenoid products. By comparison with traditional methods, microorganisms have the advantage of fast growth without the need for land to sustain development. Many microorganisms use either the methylerythritol 4-phosphate (MEP) pathway or the melavonate (MVA) pathway to supply intermediates necessary to produce terpenoid products. These MEP or MVA pathways can include an endogenous or an engineered MEP or MVA pathway or both. A detailed understanding of isoprenoid pathway engineering and optimization is disclosed in WO 2011/060057, US 2011/0189717, US 2012/107893, U.S. Pat. No. 8,512,988 and Ajikumar et al (2010) Nootkatone (4,4a,5,6,7,8-hexahydro-6-isopropenyl-4,4a-dimethyl-2(3II)-naphtalenone) is an important flavour constituent of grapefruit and is used commercially to flavour soft drinks and other beverages, as well as being used in perfumery. The conventional method for nootkatone preparation is by oxidation of valencene (see, e.g., U.S. Pat. Nos. 6,200,786 and 8,097,442). The starting material valencene is expensive and thus methods that consume valencene are less commercially acceptable. Because of these drawbacks, there is a need for commercially feasible and sustainable methods to prepare nootkatone and associated products. An object of the present disclosure is to provide sustainable production of oxygenated sesquiterpene products. Specifically, the present disclosure provides enzyme catalysts for the ex vivo or in vivo production of certain oxygenated sesquiterpenes. In some embodiments, the disclosure provides host cells engineered for the biosynthesis of the oxygenated sesquiterpenes. Another object of the present disclosure is to provide engineered cytochrome P450 (CYP450) enzymes for synthesis of oxygenated sesquiterpenes, including in some embodiments functional expression alongside a reductase counterpart in In one aspect, the disclosure provides a method for making an oxygenated product of a sesquiterpene. The method comprises contacting the sesquiterpene with In some embodiments, the method takes place in an ex vivo (e.g., cell free) system. In other embodiments, the sesquiterpene substrate and the SrKO or derivative thereof are contacted in a cell expressing the SrKO, such as a bacterium (e.g., In some embodiments, the SrKO derivative has at least one mutation with respect to the wild type SrKO that increases valencene oxidase activity, or increases production of nootkatone, α-nootkatol, and/or β-nootkatol. For example, the SrKO may have from 1 to 50 mutations independently selected from substitutions, deletions, or insertions relative to wild type SrKO (SEQ ID NOS: 37 and 108) or an SrKO modified for expression and activity in In accordance with aspects of the disclosure, oxygenated sesquiterpene products are obtainable by contacting a sesquiterpene substrate with In various embodiments, the sesquiterpene substrate is (or the predominant sesquiterpene substrate is) valencene, germacrene (A, B, C, D, or E), farnesene, farnesol, nootkatol, patchoulol, cadinene, cedrol, humulene, longifolene, and/or bergamotene, β-ylangene, β-santalol, β-santalene, α-santalene, α-santalol, β-vetivone, α-vetivone, khusimol, bisabolene, β-aryophyllene, longifolene; α-sinensal; α-bisabolol, (−)-β-copaene, (−)-α-copaene, 4(Z),7(Z)-ecadienal, cedrol, cedrene, cedrol, guaiol, (−)-6,9-guaiadiene, bulnesol, guaiol, ledene, ledol, lindestrene, and alpha-bergamotene. In some embodiments, the predominant sesquiterpene substrate is valencene, and the predominant oxygenated product is nootkatone and/or nootkatol, which in some embodiments comprises both α and β nootkatol. The disclosure, when applied in vivo, is applicable to a wide array of host cells. In some embodiments, the host cell is a microbial host, such as a bacterium selected from In some embodiments, the host cell produces isopentyl pyrophosphate (IPP), which acts as a substrate for the synthesis of the sesquiterpene. In some embodiments, the IPP is produced by metabolic flux through an endogenous or heterologous methylerythritol phosphate (MEP) or mevalonic acid (MVA) pathway. In some embodiments, the sesquiterpene is produced at least in part by metabolic flux through an MEP pathway, and wherein the host cell has at least one additional copy of a dxs, ispD, ispF, and/or idi gene. In some embodiments, the host cell expresses a farnesyl pyrophosphate synthase (FPPS), which produces farnesyl pyrophosphate (FPP) from IPP or DMAPP. The host cell may further express a heterologous sesquiterpene synthase to produce the desired sesquiterpene scaffold. For example, in some embodiments the cell expresses a valencene synthase. Several valencene synthase enzymes are known including The SrKO or derivative thereof acts on the sesquiterpene (e.g., valencene) to produce the oxygenated terpene product. In some embodiments the SrKO is a fusion protein with a cytochrome P450 reductase partner (e.g., SrCPR), allowing the cofactor to be efficiently regenerated. In other embodiments, a P450 reductase is provided (e.g., to in vitro system) or expressed in the host cell separately, and may be expressed in the same operon as the SrKO in some embodiments. In some embodiments, the CPR enzyme is expressed separately, and the gene may be integrated into the host cell genome in some embodiments. Various exemplary CPR enzymes are disclosed herein, and which may be derivatized to improve oxygenated sesquiterpenoid titer and/or to improve P450 efficiency. In some embodiments, the host cell expresses one or more enzymes that further direct oxygenated product to nootkatone, such as the expression of one or more alcohol dehydrogenase (ADH) enzymes. Exemplary ADH enzymes are disclosed herein. In other aspects, the disclosure provides a method for making a product containing an oxygenated sesquiterpene, which comprises incorporating the oxygenated sesquiterpene prepared and recovered according to the methods described herein into a consumer or industrial product. For example, the product may be a flavor product, a fragrance product, a cosmetic, a cleaning product, a detergent or soap, or a pest control product. In some embodiments, the oxygenated product recovered comprises nootkatol (e.g., α and/or β nootkatol) and/or nootkatone, and the product is a flavor product selected from a beverage, a chewing gum, a candy, or a flavor additive. In other aspects, the disclosure provides engineered SrKO enzymes having enhanced valencene oxidase activity as compared to wild type, as well as host cells producing an oxygenated sesquiterpene as described herein, and which express all of the enzyme components for producing the desired oxygenated sesquiterpene from isopentyl pyrophosphate (IPP). For example, the host cell in various embodiments expresses a farnesyl pyrophosphate synthase, a sesquiterpene synthase, and the SrKO or derivative thereof. IPP may be produced through the MEP and/or MVA pathway, which may be endogenous to the host cell, and which may be enhanced through expression of heterologous enzymes or duplication of certain enzymes in the pathway. Host cells include various bacteria and yeast as described herein. The oxygenated sesquiterpene (e.g., nootkatone and/or nootkatol) may be recovered from the culture, and/or optionally may act as the substrate for further chemical transformation in the cell or ex vivo system. In another aspect, the disclosure provides sesquiterpene-containing oil produced by the methods and host cells described herein. In some embodiments, the oil comprises hydroxygermacra-1(10)5-diene, murolan-3,9(11) diene-10-peroxy, alpha-nootkatol, beta-nootkatol, and nootkatone. In some embodiments, the predominant oxygenated products of valencene is nootkatone and nootkatol, and the oxygenated sesquiterpene product comprises both alpha and beta nootkatol. In another aspect, there is provided an SrKO crystal model structure (CMS) based on the structural coordinates of P45017A1 (which catalyzes the biosynthesis of androgens). The CMS, including the terpene binding pocket domain (TBD) that comprises a terpene binding pocket (TBP) and a terpene (e.g., valencene) bound to the TBD, is illustrated in Additional aspects and embodiments of the invention will be apparent from the following detailed description. The present disclosure in various aspects provides methods for making oxygenated terpenes or terpenoids in ex vivo or in cell systems. The disclosure further provides engineered or modified enzymes, polynucleotides, and host cells for use in such methods. The disclosure in various embodiments is directed to a method to produce nootkatone and/or nootkatol using an SrKO enzyme. Surprisingly, it was found that the SrKO enzyme can be used to catalyze sesquiterpene oxidation (e.g., valencene oxidation to nootkatol and nootkatone). As used herein, SrKO refers to ent-kaurene oxidase CYP701A5 [ As used herein, the term “SrKO derivative,” “modified SrKO polypeptide,” “engineered SrKO,” “SrKO variant,” “engineered valencene oxidase,” or “valencene oxidase variant” refers to an amino acid sequence that has substantial structural and/or sequence identity with SrKO, and catalyzes oxygenation of a sesquiterpene scaffold, such as valencene. SrKO enzymes engineered for the oxygentation of valencene are also referred to herein as “valencene oxidase” or “VO” enzymes. Generally, derivatives comprise mutated forms of SrKO having at least one mutation that increases the activity of the enzyme for the valencene substrate or for the production of nootkatone, nootkatol, and/or other products. Some SrKO mutations are provided in Tables 2.1, 2.2, and 2.3. Some such additional SrKO mutations are provided in Table 6. The term “contacting” means that the components are physically brought together, whether in vivo through co-expression of relevant protein products (e.g., sesquiterpene synthase and CYP450) in a host cell, or by adding or feeding a substrate of interest to a host cell expressing an SrKO or derivative thereof, or in vitro (or “ex vivo”) by adding sesquiterpene substrate to purified P450 enzyme or cellular extract or partially purified extract containing the same. The terms in vitro and ex vivo refer to a cell free system, and may be performed in a reaction tube or well. As used herein, “terpenes” are a large and varied class of hydrocarbons that have a simple unifying feature, despite their structural diversity. According to the “isoprene rule”, all terpenes consist of isoprene (C5) units. This fact is used for a rational classification depending on the number of such units. Monoterpenes comprise 2 isoprene units and are classified as (C10) terpenes, sesquiterpenes comprise 3 isoprene units and are classified as (C15) terpenes, diterpenes comprise 4 isoprene units and are classified as (C20) terpenes, sesterterpenes (C25), triterpenes (C30) and rubber (C5)n. They occur as acyclic or mono- to pentacyclic derivatives with alcohol, ether, ester, aldehyde, or ketone groups (the so called “terpenoids”), everywhere in organisms, particularly in higher plants, and are characteristic of the individual type of plants. Terpenes such as Monoterpenes (C10), Sesquiterpenes (C15) and Diterpenes (C20) are derived from the prenyl diphosphate substrates, geranyl diphosphate (GPP), farnesyl diphosphate (FPP) and geranylgeranyl diphosphate (GGPP) respectively through the action of a very large group of enzymes called the terpene (terpenoid) synthases. These enzymes are often referred to as terpene cyclases since the product of the reactions are cyclised to various monoterpene, sesquiterpene and diterpene carbon skeleton products. Many of the resulting carbon skeletons undergo subsequence oxygenation by cytochrome p450 hydrolysase enzymes to give rise to large families of derivatives. The technical syntheses of top-selling flavours and fragrances can start from terpenes which can also serve as excellent solvents or diluting agents for dyes and varnishes. Natural or synthetic resins of terpenes are used and also many pharmaceutical syntheses of vitamins and insecticides start from terpenes. As used herein, the term “terpene” or “sesquiterpene” (for example) includes corresponding terpenoid or sesquiterpenoid compounds. As used herein, the term “oxygenated sesquiterpene” refers to a sesquiterpene scaffold having one or more oxygenation events, producing a corresponding alcohol, aldehyde, carboxylic acid and/or ketone. One or more oxygenated sesquiterpenes may be referred to herein as an “oxygenated product.” As used herein, the term “unoxygenated sesquiterpene” refers to a sesquiterpene scaffold that has not undergone any oxygenation events. An unoxygenated sesquiterpene may also be referred to herein as an “unoxygenated product.” As used herein, the term “oxygenated product titer” or “oxygenated titer” refers to the sum of titers of α-nootkatol, β-nootkatol, and (+)-nootkatone. As used herein, the term “MEP pathway” refers to the (2-C-methyl-D-erythritol 4-phosphate) pathway, also called the MEP/DOXP (2-C-methyl-D-erythritol 4-phosphate/1-deoxy-D-xylulose 5-phosphate) pathway or the non-mevalonate pathway or the mevalonic acid-independent pathway. In the MEP pathway, pyruvate and D-glyceraldehyde-3-phosphate are converted via a series of reactions to IPP and DMAPP. The pathway typically involves action of the following enzymes: 1-deoxy-D-xylulose-5-phosphate synthase (Dxs), 1-deoxy-D-xylulose-5-phosphate reductoisomerase (IspC), 4-diphosphocytidyl-2-C-methyl-D-erythritol synthase (IspD), 4-diphosphocytidyl-2-C-methyl-D-erythritol kinase (IspE), 2C-methyl-D-erythritol 2,4-cyclodiphosphate synthase (IspF), 1-hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate synthase (IspG), and isopentenyl diphosphate isomerase (IspH). The MEP pathway, and the genes and enzymes that make up the MEP pathway, are described in U.S. Pat. No. 8,512,988, which is hereby incorporated by reference in its entirety. For example, genes that make up the MEP pathway include dxs, ispC, ispD, ispE, ispF, ispG, ispH, idi, and ispA. As used herein, the “MVA pathway” refers to the biosynthetic pathway that converts acetyl-CoA to IPP. The mevalonate pathway typically comprises enzymes that catalyze the following steps: (a) condensing two molecules of acetyl-CoA to acetoacetyl-CoA (e.g., by action of acetoacetyl-CoA thiolase); (b) condensing acetoacetyl-CoA with acetyl-CoA to form hydroxymethylglutaryl-CoenzymeA (HMG-CoA) (e.g., by action of HMG-CoA synthase (HMGS)); (c) converting HMG-CoA to mevalonate (e.g., by action of HMG-CoA reductase (HMGR)); (d) phosphorylating mevalonate to mevalonate 5-phosphate (e.g., by action of mevalonate kinase (MK)); (e) converting mevalonate 5-phosphate to mevalonate 5-pyrophosphate (e.g., by action of phosphomevalonate kinase (PMK)); and (f) converting mevalonate 5-pyrophosphate to isopentenyl pyrophosphate (e.g., by action of mevalonate pyrophosphate decarboxylase (MPD)). The MVA pathway, and the genes and enzymes that make up the MEP pathway, are described in U.S. Pat. No. 7,667,017, which is hereby incorporated by reference in its entirety. As used herein, the term “cytochrome P450 reductase partner” or “CPR partner” refers to a cytochrome P450 reductase capable of regenerating the cofactor component of the cytochrome P450 oxidase of interest (e.g., SrKO) for oxidative chemistry. For example, SrCPR is a natural CPR partner for SrKO. In some embodiments, the CPR partner is not the natural CPR partner for SrKO. In some embodiments employing in vivo production of oxygenated sesquiterpene, the SrKO and SrCPR are co-expressed as separate proteins, or in some embodiments are expressed as a fusion protein. As used herein, the term “natural product” refers to a product obtained, at least in part, from plant and/or animal material or obtained from microbial enzymatic biotransformations/bioconversions/biocatalysis and/or biosynthesis. Ranges can be expressed herein as from “about” one particular value, and/or to “about” another particular value. When such a range is expressed, another embodiment includes from the one particular value and/or to the other particular value. Similarly, when values are expressed as approximations, by use of the antecedent “about,” it will be understood that the particular value forms another embodiment. It will be further understood that the endpoints of each of the ranges are significant both in relation to the other endpoint, and independently of the other endpoint. It is also understood that there are a number of values disclosed herein, and that each value is also herein disclosed as “about” that particular value in addition to the value itself. For example, if the value “10” is disclosed, then “about 10” is also disclosed. It is also understood that each unit between two particular units are also disclosed. For example, if 10 and 15 are disclosed, then 11, 12, 13, and 14 are also disclosed. The similarity of nucleotide and amino acid sequences, i.e., the percentage of sequence identity, can be determined via sequence alignments. Such alignments can be carried out with several art-known algorithms, such as with the mathematical algorithm of Karlin and Altschul (Karlin & Altschul (1993) Proc. Natl. Acad. Sci. USA 90: 5873-5877), with hmmalign (HMMER package, hmmer.wustl.edu/) or with the CLUSTAL algorithm (Thompson, J. D., Higgins, D. G. & Gibson, T. J. (1994) Nucleic Acids Res. 22, 4673-80). The grade of sequence identity (sequence matching) may be calculated using e.g. BLAST, BLAT or BlastZ (or BlastX). A similar algorithm is incorporated into the BLASTN and BLASTP programs of Altschul et al (1990) J. Mol. Biol. 215: 403-410. BLAST polynucleotide searches can be performed with the BLASTN program, score=100, word length=12. BLAST protein searches may be performed with the BLASTP program, score=50, word length=3. To obtain gapped alignments for comparative purposes, Gapped BLAST is utilized as described in Altschul et al (1997) Nucleic Acids Res. 25: 3389-3402. When utilizing BLAST and Gapped BLAST programs, the default parameters of the respective programs are used. Sequence matching analysis may be supplemented by established homology mapping techniques like Shuffle-LAGAN (Brudno M., Bioinformatics 2003b, 19 Suppl 1:154-162) or Markov random fields. “Conservative substitutions” may be made, for instance, on the basis of similarity in polarity, charge, size, solubility, hydrophobicity, hydrophilicity, and/or the amphipathic nature of the amino acid residues involved. The 20 naturally occurring amino acids can be grouped into the following six standard amino acid groups: (1) hydrophobic: Met, Ala, Val, Leu, He; (2) neutral hydrophilic: Cys, Ser, Thr; Asn, Gin; (3) acidic: Asp, Glu; (4) basic: His, Lys, Arg; (5) residues that influence chain orientation: Gly, Pro; and (6) aromatic: Trp, Tyr, Phe. As used herein, “conservative substitutions” are defined as exchanges of an amino acid by another amino acid listed within the same group of the six standard amino acid groups shown above. For example, the exchange of Asp by Glu retains one negative charge in the so modified polypeptide. In addition, glycine and proline may be substituted for one another based on their ability to disrupt a-helices. Some preferred conservative substitutions within the above six groups are exchanges within the following sub-groups: (i) Ala, Val, Leu and He; (ii) Ser and Thr; (ii) Asn and Gin; (iv) Lys and Arg; and (v) Tyr and Phe. As used herein, “non-conservative substitutions” or “non-conservative amino acid exchanges” are defined as exchanges of an amino acid by another amino acid listed in a different group of the six standard amino acid groups (1) to (6) shown above. In one aspect, the disclosure provides a method for making an oxygenated product of a sesquiterpene. In various embodiments, the sesquiterpene substrate is (or the predominant sesquiterpene substrate is) valencene, germacrene (A, B, C, D, or E), farnesene, farnesol, nootkatol, patchoulol, cadinene, cedrol, humulene, longifolene, and/or bergamotene, β-ylangene, β-santalol, β-santalene, α-santalene, α-santalol, β-vetivone, α-vetivone, khusimol, bisabolene, β-aryophyllene, Longifolene; α-sinensal; α-bisabolol, (−)-β-copaene, (−)-α-copaene, 4(Z),7(Z)-ecadienal, cedrol, cedrene, cedrol, guaiol, (−)-6,9-guaiadiene, bulnesol, guaiol, ledene, ledol, lindestrene, and alpha-bergamotene. In some embodiments, the predominant sesquiterpene substrate is valencene, and the predominant oxygenated product is nootkatone and/or nootkatol, which in some embodiments comprises both α and β nootkatol. In this context, the term “predominant” means that the particular sesquiterpene is present at a level higher than all other terpene or terpenoid species individually. In some embodiments, the predominant sesquiterpene (either the substrate or the oxygenated product after the reaction) makes up at least 25%, at least 40%, at least 50%, or at least 75% of the terpene or terpenoid component of the composition. In various embodiments involving in vivo production of oxygenated sesquiterpenes, the oxygenated product is recovered from the culture media, and can be fractionated to isolate or enrich for various components of the product, such as nootkatone, nootkatol, and/or other components. In various embodiments, the disclosure comprises contacting a sesquiterpene with a terpene oxidizing P450 enzyme, or a derivative thereof. The contacting may take place in a host cell or in a cell free system. The substrate for oxidation (e.g., the sesquiterpene), may be produced by the cells (e.g., through metabolic flux through the MEP or MVA pathways), or alternatively fed to the host cells expressing the P450 enzyme. The oxygenated product may be recovered, or be the substrate for further chemical transformation either in the cellular system or cell free system. Table 1 below provides a list of exemplary P450 enzymes. While in certain embodiments the disclosure involves the use of the following P450 enzymes (optionally engineered to increase the oxygenation of valencene to nootkatone and/or nootkatol), a preferred enzyme in accordance with this disclosure is SrKO. Exemplary oxygenated sesquiterpene products obtained by these reactions in accordance with the disclosure are shown in Table 4. In various embodiments, the method comprises contacting the sesquiterpene with a protein comprising In some embodiments, the SrKO derivative comprises an amino acid sequence that has from about 1 to about 50 mutations independently selected from substitutions, deletions, or insertions relative to SrKO (SEQ ID NO: 37 or 38), or relative to an SrKO enzyme modified at its N-terminus for functional expression in In these or other embodiments, the SrKO derivative may comprise an amino acid sequence having at least about 50% sequence identity, at least about 55% sequence identity, at least about 60% sequence identity, at least about 65% sequence identity, at least about 70% sequence identity, at least about 75% sequence identity, at least about 80% sequence identity, at least about 85% sequence identity, or at least 90% sequence identity, or at least 91% sequence identity, or at least 92% sequence identity, or at least 93% sequence identity, or at least 94% sequence identity, or at least 95% sequence identity, or at least 96% sequence identity, or at least 97% sequence identity, or at least 98% sequence identity, or at least 99% sequence identity, to SrKO (SEQ ID NO: 37, 38, or 55). In various embodiments, the SrKO derivative has higher activity for the oxygenation of valencene than the wild type enzyme, such as a higher production of oxygenated oil upon contact with valencene substrate than the wild type enzyme (SEQ ID NO: 37) or the wild type enzyme as modified for functional expression in In some embodiments, mutants are selected for an increase in production of oxygenated valencene, such as nootkatone, α-nootkatol, and/or β-nootkatol. For example, the SrKO derivative may have one or more mutations at positions selected from 46, 76, 94, 131, 231, 284, 383, 390, 400, 444, 468, 488 and 499, numbered according to SEQ ID NO: 37. For example, in some embodiments, the SrKO is a derivative comprising an amino acid sequence having one or more (or all) of the mutations selected from H46R, R76K, M94V, T131Q, F231L, H284Q, R383K, I390L, V400Q, I444A, T468I, T488D, and T499N, numbered according to SEQ ID NO: 37. In certain embodiments, the SrKO is a derivative comprising an amino acid sequence having one or more (or all) of the mutations selected from R76K, M94V, T131Q, F231L, H284Q, R383K, I390L, T468I, and T499N, numbered according to SEQ ID NO: 37. In some embodiments, the SrKO derivative comprises an amino acid sequence selected from SEQ ID NOS: 55-61, which were engineered according to this disclosure to improve activity for oxygenation of valencene (e.g., production of nootkatone). In some embodiments, the derivative comprises an amino acid sequence having from one to twenty mutations relative to a sequence selected from SEQ ID NOS: 55-61, with the proviso that the amino acid sequence has one or more mutations at positions selected from 46, 76, 94, 131, 231, 284, 383, 390, 400, 444, 468, 488, and 499 (numbered according to SEQ ID NO: 37), or the proviso that the SrKO derivative comprises an amino acid sequence having one or more (or all) of the mutations selected from H46R, R76K, M94V, T131Q, F231L, H284Q, R383K, I390L, V400Q, I444A, T468I, T488D, and T499N (numbered according to SEQ ID NO: 37). In certain embodiments, the derivative comprises an amino acid sequence having from one to twenty mutations relative to a sequence selected from SEQ ID NOS: 55-61, with the proviso that the amino acid sequence has one or more mutations at positions selected from 46, 76, 94, 131, 231, 284, 383, 390, 400, 444, 468, 488, and 499 (numbered according to SEQ ID NO: 37), or the proviso that the SrKO derivative comprises an amino acid sequence having one or more (or all) of the mutations selected from R76K, M94V, T131Q, F231L, H284Q, R383K, I390L, T468I, and T499N (numbered according to SEQ ID NO: 37). In some embodiments, the disclosure provides a recombinant polynucleotide encoding the SrKO derivative described above, which may be inserted into expression vectors for expression and optional purification. In some embodiments, the polynucleotide is incorporated into the genome of valencene-producing cells, such as valencene-producing The SrKO or derivative in various embodiments has valencene oxidase activity. Assays for determining and quantifying valencene oxidase activity are described herein and are known in the art. Assays include expressing the SrKO (or derivative) in valencene-producing cells (e.g., The SrKO may be expressed in a variety of host cells, either for recombinant protein production, or for sesquiterpene (e.g., valencene) oxidation. For example, the host cells include those described in U.S. Pat. No. 8,512,988, which is hereby incorporated by reference in its entirety. The host cell may be a prokaryotic or eukaryotic cell. In some embodiments the cell is a bacterial cell, such as In some embodiments, the host cell produces isopentyl pyrophosphate (IPP), which acts as a substrate for the synthesis of the sesquiterpene. In some embodiments, the IPP is produced by metabolic flux (e.g., starting with a carbon source supplied to the cell) through an endogenous or heterologous methylerythritol phosphate (MEP) or mevalonic acid (MVA) pathway. In certain embodiments, the MEP or MVA pathway may be enhanced through expression of heterologous enzymes or duplication of certain enzymes in the pathway. The MEP (2-C-methyl-D-erythritol 4-phosphate) pathway, also called the MEP/DOXP (2-C-methyl-D-erythritol 4-phosphate/1-deoxy-D-xylulose 5-phosphate) pathway or the non-mevalonate pathway or the mevalonic acid-independent pathway refers to the pathway that converts glyceraldehyde-3-phosphate and pyruvate to IPP and DMAPP. The pathway typically involves action of the following enzymes: 1-deoxy-D-xylulose-5-phosphate synthase (Dxs), 1-deoxy-D-xylulose-5-phosphate reductoisomerase (IspC), 4-diphosphocytidyl-2-C-methyl-D-erythritol synthase (IspD), 4-diphosphocytidyl-2-C-methyl-D-erythritol kinase (IspE), 2C-methyl-D-erythritol 2,4-cyclodiphosphate synthase (IspF), 1-hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate synthase (IspG), and isopentenyl diphosphate isomerase (IspH). The MEP pathway, and the genes and enzymes that make up the MEP pathway, are described in U.S. Pat. No. 8,512,988, which is hereby incorporated by reference in its entirety. For example, genes that make up the MEP pathway include dxs, ispC, ispD, ispE, ispF, ispG, ispH, idi, and ispA. In some embodiments, the sesquiterpene is produced at least in part by metabolic flux through an MEP pathway, and wherein the host cell has at least one additional copy of a dxs, ispD, ispF, and/or idi gene (e.g., dxs and idi; or dxs, ispD, ispF, and/or idi). The MVA pathway refers to the biosynthetic pathway that converts acetyl-CoA to IPP. The mevalonate pathway typically comprises enzymes that catalyze the following steps: (a) condensing two molecules of acetyl-CoA to acetoacetyl-CoA (e.g., by action of acetoacetyl-CoA thiolase); (b) condensing acetoacetyl-CoA with acetyl-CoA to form hydroxymethylglutaryl-CoenzymeA (HMG-CoA) (e.g., by action of HMG-CoA synthase (HMGS)); (c) converting HMG-CoA to mevalonate (e.g., by action of HMG-CoA reductase (HMGR)); (d) phosphorylating mevalonate to mevalonate 5-phosphate (e.g., by action of mevalonate kinase (MK)); (e) converting mevalonate 5-phosphate to mevalonate 5-pyrophosphate (e.g., by action of phosphomevalonate kinase (PMK)); and (f) converting mevalonate 5-pyrophosphate to isopentenyl pyrophosphate (e.g., by action of mevalonate pyrophosphate decarboxylase (MPD)). The MVA pathway, and the genes and enzymes that make up the MEP pathway, are described in U.S. Pat. No. 7,667,017, which is hereby incorporated by reference in its entirety. In some embodiments, the host cell expresses a farnesyl pyrophosphate synthase (FPPS), which produces farnesyl pyrophosphate from IPP or DMAPP. As shown in The host cell may further express a heterologous sesquiterpene synthase to produce the desired sesquiterpene, such as a valencene synthase. Several valencene synthase enzymes are known including valencene synthase from For example, in some embodiments, the valencene synthase is a VvVS derivative that comprises an amino acid sequence having from about 1 to about 40 mutations, from about 1 to about 35 mutations, from about 1 to about 30 mutations, about 1 to about 25 mutations, from about 1 to about 20 mutations, about 1 to about 15 mutations, or from about 1 to about 10 mutations independently selected from substitutions, deletions, or insertions with respect to VvVS (SEQ ID NO: 1). For example, the VvVS derivative may comprise an amino acid sequence having at least about 5 or at least about 10, but less than about 30 or about 20 mutations with respect to SEQ ID NO: 1. In various embodiments, the VvVS derivative comprises an amino acid sequence that has about 1 mutation, about 2 mutations, about 3 mutations, about 4 mutations, about 5 mutations, about 6 mutations, about 7 mutations, about 8 mutations, about 9 mutations, about 10 mutations, about 11 mutations, about 12 mutations, about 13 mutations, about 14 mutations, about 15 mutations, about 16 mutations, about 17 mutations, about 18 mutations, about 19 mutations, about 20 mutations, about 21 mutations, about 22 mutations, about 23 mutations, about 24 mutations, about 25 mutations, about 26 mutations, about 27 mutations, about 28 mutations, about 29 mutations, about 30 mutations, about 31 mutations, about 32 mutations, about 33 mutations, about 34 mutations, about 35 mutations, about 36 mutations, about 37 mutations, about 38 mutations, about 39 mutations, or about 40 mutations relative to VvVS (SEQ ID NO: 1). Such sequences may have at least 60% sequence identity, at least 70% sequence identity, at least 80% sequence identity, at least 90% sequence identity, at least 95% sequence identity, or at least about 96%, about 97%, about 98%, or about 99% sequence identity with SEQ ID NO:1. Exemplary mutations of VvVS are shown in Table 3. Mutations can be guided by a homology model of Thus, in various embodiments, the engineered VvVS may have at least about 1 mutation, about 2 mutations, about 3 mutations, about 4 mutations, about 5 mutations, about 6 mutations, about 7 mutations, about 8 mutations, about 9 mutations, about 10 mutations, about 11 mutations, about 12 mutations, about 13 mutations, about 14 mutations, about 15 mutations, about 16 mutations, about 17 mutations, about 18 mutations, about 19 mutations, about 20 mutations, about 21 mutations, about 22 mutations, about 23 mutations, about 24 mutations, about 25 mutations, about 26 mutations, about 27 mutations, about 28 mutations, about 29 mutations, about 30 mutations, about 31 mutations, about 32 mutations, about 33 mutations, about 34 mutations, about 35 mutations, about 36 mutations, about 37 mutations, about 38 mutations, about 39 mutations, or about 40 mutations selected from Table 3. Exemplary recombinant valencene synthases Vv1M1 (SEQ ID NO: 3), Vv2M1 (SEQ ID NO: 5), Vv1M5 (SEQ ID NO: 7), Vv2M5 (SEQ ID NO: 9), and VS2 (SEQ ID NO: 11) are further depicted in In certain aspects, the disclosure provides polynucleotides comprising a nucleotide sequence encoding a valencene synthase modified for increased expression of valencene as described above. Such polynucleotides may be expressed in host cells, either on extrachromosomal elements such as plasmids, or may be chromosomally integrated. In various embodiments, the SrKO is expressed alongside a P450 reductase to regenerate the enzyme, or alternatively, the SrKO or derivative is expressed with the P450 reductase as a chimeric P450 enzyme. Functional expression of cytochrome P450 has been considered challenging due to the inherent limitations of bacterial platforms, such as the absence of electron transfer machinery and cytochrome P450 reductases, and translational incompatibility of the membrane signal modules of P450 enzymes due to the lack of an endoplasmic reticulum. Accordingly, in some embodiments the SrKO is expressed as a fusion protein with a cytochrome P450 reductase partner. Cytochrome P450 reductase is a membrane protein found in the endoplasmic reticulum. It catalyzes pyridine nucleotide dehydration and electron transfer to membrane bound cytochrome P450s. Isozymes of similar structure are found in humans, plants, other mammals, and insects. Exemplary P450 reductase partners include, for example, Engineering of P450 fusion proteins is disclosed, for example, in US 2012/0107893 and US 2012/0164678, both of which are hereby incorporated by reference in their entireties. In certain embodiments, the SrKO is fused to the cytochrome P450 reductase partner through a linker. Exemplary linker sequences, which are predominantly serine, glycine, and/or alanine, and optionally from one to five charged amino acids such as lysine or arginine, include, for example, GSG, GSGGGGS (SEQ ID NO: 113), GSGEAAAK (SEQ ID NO: 114), GSGEAAAKEAAAK (SEQ ID NO: 115), GSGMGSSSN (SEQ ID NO: 116), and GSTGS (SEQ ID NO: 117). The linker is generally flexible, and contains no more than one, two, or three hydrophobic residues, and is generally from three to fifty amino acids in length, such as from three to twenty amino acids in length. In other embodiments, a P450 reductase is expressed in the host cell separately, and may be expressed in the same operon as the SrKO in some embodiments. In some embodiments, the P450 reductase enzyme is expressed separately in the host cell, and the gene is optionally integrated into the genome or expressed from a plasmid. In certain embodiments the N-terminus of the P450 enzymes may be engineered to increase their functional expression. The N-terminus of membrane-bound P450 plays important roles in enzyme expression, membrane association and substrate access. It has been reported that the use of rare codons in the N-terminus of P450 significantly improved the expression level of P450. Further, since most plant P450 enzymes are membrane-bound and hydrophobic substrates are thought to enter the enzymes through channels dynamically established between the P450 and membrane, N-terminal engineering can affect the association of the membrane and P450 and therefore the access of substrate to the enzyme. Accordingly, in an embodiment, N-terminal engineering of SrKO generates an SrKO derivative that either maintains or shows enhanced valencene oxidase activity in a host system such as In some embodiments, the SrKO is a derivative having a deletion of at least a portion of its N-terminal transmembrane region, and the addition of an inner membrane transmembrane domain from In some embodiments, the host cell further expresses one or more enzymes, such as an alcohol dehydrogenase (ADH). In certain embodiments, the host cell may express an ADH enzyme producing nootkatone from nootkatol, examples of which include Sesquiterpenes (e.g., valencene and its oxygenated products) can be produced as biosynthetic products of the non-mevalonate pathway in WO 2011/060057, US 2011/0189717, US 2012/107893, and U.S. Pat. No. 8,512,988 (each of which are hereby incorporated by reference) describe methods and compositions for optimizing production of terpenoids in cells by controlling expression of genes or proteins participating in an upstream pathway and a downstream pathway. This can be achieved by grouping the enzyme pathways into two modules: an upstream (MEP) pathway module (e.g., containing one or more genes of the MEP pathway) and a downstream, heterologous pathway to sesquiterpene production. Using this basic configuration, parameters such as the effect of plasmid copy number on cell physiology, gene order and promoter strength in an expression cassette, and chromosomal integration are evaluated with respect to their effect on terpene and terpenoid (e.g., sesquiterpene) production. Expression of genes within the MEP pathway can thus be regulated in a modular method. As used herein, regulation by a modular method refers to regulation of multiple genes together. By way of example, multiple genes within the MEP pathway can be recombinantly expressed on a contiguous region of DNA, such as an operon. It should be appreciated that modules of genes within the MEP pathway, consistent with aspects of the disclosure, can contain any of the genes within the MEP pathway, in any order. In some embodiments, a gene within the MEP pathway is one of the following: dxs, ispC, ispD, ispE, ispF, ispG, ispH, idi, ispA or ispB. A non-limiting example of a module of genes within the MEP pathway is a module containing the genes dxs, idi, ispD and ispF, and referred to as dxs-idi-ispDF. The manipulation of the expression of genes and/or proteins, including modules such as the dxs-idi-ispDF operon, and a FPPS-VS operon, can be achieved through methods known to one of ordinary skill in the art. For example, expression of the genes or operons can be regulated through selection of promoters, such as inducible promoters, with different strengths. Several non-limiting examples of promoters include Trc, T5 and T7. Additionally, expression of genes or operons can be regulated through manipulation of the copy number of the gene or operon in the cell. The expression of one or more genes and/or proteins within the MEP pathway can be upregulated and/or downregulated. In certain embodiments, upregulation of one or more genes and/or proteins within the MEP pathway can be combined with downregulation of one or more genes and/or proteins within the MEP pathway. By way of example, in some embodiments, a cell that overexpresses one or more components of the non-mevalonate (MEP) pathway is used, at least in part, to amplify isopentyl diphosphate (IPP) and dimethylallyl diphosphate (DMAPP), substrates of GGPPS. In some embodiments, overexpression of one or more components of the non-mevalonate (MEP) pathway is achieved by increasing the copy number of one or more components of the non-mevalonate (MEP) pathway. In this regards, copy numbers of components at rate-limiting steps in the MEP pathway such as (dxs, ispD, ispF, idi) can be amplified, such as by additional episomal expression. In some embodiments, the production of indole is used as a surrogate marker for sesquiterpene production, and/or the accumulation of indole in the culture is controlled to increase sesquiterpene production. For example, in various embodiments, accumulation of indole in the culture is controlled to below about 100 mg/L, or below about 75 mg/L, or below about 50 mg/L, or below about 25 mg/L, or below about 10 mg/L. The accumulation of indole can be controlled by balancing protein expression and activity using the multivariate modular approach described above, and/or is controlled by chemical means. In other aspects, the disclosure provides a method for making a product containing an oxygenated sesquiterpene (as described), which comprises incorporating the oxygenated sesquiterpene prepared and recovered according to the method described above into a consumer or industrial product. For example, the product may be a flavor product, a fragrance product, a cosmetic, a cleaning product, a detergent or soap, or a pest control product (e.g., an insect repellant). In some embodiments, the oxygenated product recovered and optionally enriched by fractionation is nootkatol (e.g., a and R nootkatol) and/or nootkatone, and the product is a flavor product selected from a beverage, a chewing gum, a candy, or a flavor additive, or is a pest control product (e.g., an insect repellant). The oxygenated product can be recovered by any suitable process, including partitioning the desired product into an organic phase. The production of the desired product can be determined and/or quantified, for example, by gas chromatography (e.g., GC-MS). The desired product can be produced in batch or continuous bioreactor systems. Production of product, recovery, and/or analysis of the product can be done as described in US 2012/0246767, which is hereby incorporated by reference in its entirety. For example, in some embodiments, oxygenated oil is extracted from aqueous reaction medium, which may be done by using an organic solvent, such as an alkane such as heptane, followed by fractional distillation. Sesquiterpene and sesquiterpenoid components of fractions may be measured quantitatively by GC/MS, followed by blending of the fractions to generate a desired nootkatone-containing ingredient for flavour (or other) applications. In other aspects, the disclosure provides polynucleotides comprising a nucleotide sequence encoding a P450 derivative described herein. The polynucleotide may be codon optimized for expression in In other aspects, the disclosure provides host cells producing an oxygenated sesquiterpene as described herein, and which express all of the enzyme components for producing the desired oxygenated sesquiterpene from isopentyl pyrophosphate (IPP). For example, the host cell in various embodiments expresses a farnesyl pyrophosphate synthase, a sesquiterpene synthase, and the SrKO or derivative thereof. IPP may be produced through the MEP and/or MVA pathway, which may be endogenous to the host cell or modified through expression of heterologous enzymes or duplication of certain enzymes in the pathway. Host cells include various bacteria and yeast as described herein. In still other aspects, the disclosure provides sesquiterpene products produced by the methods and host cells described herein. As disclosed herein, SrKO enzyme showed unique activities by creating different stereoisomers of the hydroxylated product (alpha and beta nootkatol and further oxidizing to ketone, nootkatone), and produced different oxygenated terpene products including hydroxygermacra-1(10)5-diene, and murolan-3,9(11) diene-10-peroxy. This activity provides for the incorporation of a unique valencene oxidation profile into an oil suitable for flavouring applications. In certain embodiments, the processes and methods disclosed herein provide compositions and formulations comprising an oxygenated product. In some embodiments, said compositions and formulations may further comprise an unoxygenated product, a sesquiterpene, valencene, a non-sesquiterpene component, and/or one or more additional ingredients. In particular embodiments, the compositions and formulations disclosed herein comprise at least one of valencene, hydroxygermacra-1(10)5-diene, murolan-3,9(11) diene-10-peroxy, α-nootkatol, β-nootkatol, and nootkatone. In certain embodiments of the compositions and formulations disclosed herein, the compositions and formulations comprise at least one of nootkatone, α-nootkatol, β-nootkatol, and valencene. For example, in certain such embodiments, the nootkatone content may be selected from about 50% to about 65% (w/w), about 52.5% to about 62.5% (w/w), and about 55% to about 60% (w/w); the α-nootkatol content may be selected from about 15% to about 30% (w/w), about 17.5% to about 27.5% (w/w), and about 20% to about 25% (w/w); the β-nootkatol content may be selected from about 1% to about 15% (w/w), about 3% to about 12% (w/w), and about 5% to about 10% (w/w); and the valencene content may be selected from about 1% to about 15% (w/w), about 3% to about 12% (w/w), and about 5% to about 10% (w/w). In specific embodiments of the compositions and formulations disclosed herein, the compositions and formulations comprise at least one of nootkatone, α-nootkatol, and β-nootkatol. For example, in certain such embodiments, the nootkatone content may be selected from about 50% to about 65% (w/w), about 52.5% to about 62.5% (w/w), and about 55% to about 60% (w/w); the α-nootkatol content may be selected from about 15% to about 30% (w/w), about 17.5% to about 27.5% (w/w), and about 20% to about 25% (w/w); and the 3-nootkatol content may be selected from about 1% to about 15% (w/w), about 3% to about 12% (w/w), and about 5% to about 10% (w/w). In one embodiment, the composition or formulation comprise nootkatone, α-nootkatol, and β-nootkatol, wherein the nootkatone is present in an amount ranging from about 55% to about 60% (w/w), the α-nootkatol is present in an amount ranging from about 20% to about 25% (w/w), and the β-nootkatol is present in an amount ranging from about 5% to about 10% (w/w). In another embodiment, the composition or formulation comprise valencene, nootkatone, α-nootkatol, and β-nootkatol, wherein the valencene is present in an amount ranging from about 5% to about 10% (w/w), the nootkatone is present in an amount ranging from about 55% to about 60% (w/w), the α-nootkatol is present in an amount ranging from about 20% to about 25% (w/w), and the β-nootkatol is present in an amount ranging from about 5% to about 10% (w/w). Further, other P450 enzymes tested, including previously known sesquiterpene CYP450's or P450's having hydroxylating activity on the valencene substrate produced one of the stereoisomers (beta nootkatol) and only minor amounts of the ketone (nootkatone). Specifically, the other sesquiterpene CYP450 enzymes produced beta-nootkatol and hydroxyl valencene as major products, while Taxol CYP450 enzyme did not produce any oxygenated valencene (Table 4 and In certain aspects, the disclosure relates to SrKO derivative enzymes. In certain embodiments, the SrKO derivative polypeptide comprises an amino acid sequence that has up to 25 mutations compared to the wild type protein according to SEQ ID NO: 37. For example, the SrKO derivative may comprise an amino acid sequence that has one or more mutations at positions selected from 46, 76, 94, 131, 231, 284, 383, 390, 400, 444, 468, 488 and 499, numbered according to SEQ ID NO: 37. For example, in some embodiments, the SrKO is a derivative comprising an amino acid sequence having one or more (or all) of the mutations selected from H46R, R76K, M94V, T131Q, F231L, H284Q, R383K, I390L, V400Q, I444A, T468I, T488D, and T499N, numbered according to SEQ ID NO:37. In certain embodiments, the SrKO is a derivative comprising an amino acid sequence having one or more (or all) of the mutations selected from R76K, M94V, T131Q, F231L, H284Q, R383K, I390L, T468I, and T499N, numbered according to SEQ ID NO: 37. In some embodiments, the SrKO derivative comprises an amino acid sequence selected from SEQ ID NOS: 55-61, which were engineered according to this disclosure to improve activity for oxygenation of valencene (e.g., production of nootkatone and/or nootkatol). In some embodiments, the derivative comprises an amino acid sequence having from one to twenty mutations relative to a sequence selected from SEQ ID NOS: 55-61, with the proviso that the amino acid sequence has one or more mutations at positions selected from 46, 76, 94, 131, 231, 284, 383, 390, 400, 444, 468, 488 and 499 (numbered according to SEQ ID NO: 37), or the proviso that the SrKO derivative comprises an amino acid sequence having one or more (or all) of the mutations selected from H46R, R76K, M94V, T131Q, F231L, H284Q, R383K, I390L, V400Q, I444A, T468I, T488D, and T499N (numbered according to SEQ ID NO: 37). In certain embodiments, the derivative comprises an amino acid sequence having from one to twenty mutations relative to a sequence selected from SEQ ID NOS: 55-61, with the proviso that the amino acid sequence has one or more mutations at positions selected from 46, 76, 94, 131, 231, 284, 383, 390, 400, 444, 468, 488 and 499 (numbered according to SEQ ID NO: 37), or the proviso that the SrKO derivative comprises an amino acid sequence having one or more (or all) of the mutations selected from R76K, M94V, T131Q, F231L, H284Q, R383K, I390L, T468I, and T499N (numbered according to SEQ ID NO: 37). As shown herein, these mutations increase the level of SrKOs valencene oxidation activity. In these or other embodiments, the SrKO is a derivative having a deletion of at least a portion of its N-terminal transmembrane region, and the addition of an inner membrane transmembrane domain from In still other aspects, the disclosure provides a method of preparing the modified SrKO polypeptide, wherein the method comprises the steps of: (i) culturing a host cell expressing the modified polypeptide under conditions which permit expression of the polypeptide; and (ii) optionally recovering the polypeptide. In still other aspects, the disclosure provides a method of producing an oxygenated sesquiterpene comprising the steps of: (i) providing the modified SrKO polypeptide, (ii) contacting a sesquiterpene with the modified SrKO polypeptide, and (iii) recovering the produced oxygenated sesquiterpene. The method may further comprise providing a CPR enzyme for regenerating the SrKO cofactor (e.g., SrCPR). In some embodiments, the oxygenated sesquiterpene is recovered as an oil. In some embodiments, the sesquiterpene is valencene. In some embodiments, the oxygenated sesquiterpene comprises hydroxygermacra-1(10)5-diene, murolan-3,9(11) diene-10-peroxy, alpha-nootkatol, beta-nootkatol, and nootkatone. In some embodiments, the predominant oxygenated product is nootkatone and/or nootkatol. In some embodiments, the oxygenated product comprises both alpha and beta nootkatol. In another aspect, there is provided an SrKO crystal model structure (CMS) based on the structural coordinates of P45017A1, with an amino acid sequence of SrKO or derivative described herein. The CMS comprises a terpene binding pocket domain (TBD) that comprises a terpene binding pocket (TBP) and a terpene (e.g., valencene) bound to the TBD. Thus, in still other embodiments, the disclosure provides a method of screening for a terpene capable of binding to a TBD wherein the method comprises the use of the SrKO CMS. In another aspect, the disclosure provides a method for screening for a terpene capable of binding to the TBP, and the method comprises contacting the TBP with a test compound, and determining if said test compound binds to said TBP. In some embodiments, the method is to screen for a test compound (e.g., terpenes) useful in modulating the activity of a SrKO enzyme. In another aspect, the disclosure provides a method for predicting, simulating or modelling the molecular characteristics and/or molecular interactions of a terpene binding domain (TBD) comprising the use of a computer model, said computer model comprising, using or depicting the structural coordinates of a terpene binding domain as defined above to provide an image of said ligand binding domain and to optionally display said image. Further, multivariate modular metabolic engineering (MMME) was applied for balancing the pathway for high level production of valencene. Naturally occurring valencene synthases, such as that from A homology model for the By applying the MMME approach, a balanced upstream and downstream valencene production strain was identified incorporating a codon-optimized version of VvVS on a plasmid with a p15A origin of replication and a T7 promoter. This strain background was then used to screen designed synthase enzyme mutations. Using the aforementioned protein engineering tools we designed over 200 unique point mutations (Table 3) which were then constructed in the p15A-T7 screening plasmid using site-directed mutagenesis. Mutated enzyme variants were transformed into the screening strain, triplicate colonies were cultured in selective LB cell culture medium overnight, and then inoculated into a minimal R-medium and cultured for four days at 22° C. Cultures were extracted using methyl tert-butyl ether (MTBE) and analyzed by combined gas chromatography/mass spectrometry for productivity of valencene. Approximately one-fifth of the designed point mutations increased valencene productivity in our screening strain by at least 20% ( Valencene was used as a model system to validate the power of CYP450-based oxygenation chemistry for production terpene chemicals. The CYPP450 candidate screening was conducted using the valencene producing A set of CYP450 enzymes, from those listed in Table 4, was selected and classified for both sesqui- and diterpene oxygenation in this Once the unique activities of SrKO were identified, experiments were conducted to improve its ability to conduct its diverse oxidation of valencene. The crystal structure for SrKO has not been described. Blast search of SrKO against RCSB Protein Data Bank shows the sequence identity of SrKO to P450 enzymes with crystal structures are low (˜20%). Given the conservative folding structures of P450s regardless of its low sequence identity, state-of-the-art protein modeling tools were used to build on SrKO. The crystal structure of membrane-bound cytochrome P450 17 A1 (see DeVore N. M., Scott E. E., In addition, a Blast search of SrKO against NCBI non-redundant protein sequence library returned no orthologs with sequence identity greater than 80% (except the SrKO itself). The top hits are listed in the Table 5. Once the unique activities of SrKO were identified, experiments were conducted to improve its ability to conduct its diverse oxidation of valencene. Using the back-to-consensus mutagenesis strategy, a multiple sequence alignment of P450 enzymes was constructed including sequences (after clustering and elimination of sequences with greater than 90% identity) from a BLAST search ofthe Uniref100 database using 4 seed kaurene oxidase genes, from a BLAST search ofthe bacterial proteome using P450BM3, P45CAM, and P40eryFas seed genes, and the most closely related SrKO homologs. Based on the homology model, the multiple sequence alignment, and the literature, various point mutations and double mutations were designed and tested. These cytochrome P450 derivatives were assessed for improvements in total oxygenated terpene productivity (e.g., total of the major peaks observed by GUMS) in the in vivo testing system described above. Mutagenesis on active site positions guided by the model revealed several variants with significantly improved oxygenated products (Table 6 and Table 7 below). The product derived from oxidation of valencene by the cytochrome P450 enzyme SrKO (SEQ ID NO: 38) was analysed by GC/MS (Agilent 6800; Column: Rtx-5, 0.32 mm×60 m×1.0 μm film thickness; GC Temp. Program: 40° C. for 5 min, increased at 4° C./min to 300° C. and held for 30 min.) resulting in the data provided in Table 8A and 8B. Similar analysis was conducted on the product produced by SrKO derivatives. It was confirmed that product profiles are comparable, and that the major products of nootkatone, α-nootkatol, and β-nootkatol can be produced at higher levels based on mutagenesis of SrKO. The oxygenated oil product can then be extracted from the aqueous reaction medium using an appropriate solvent (e.g., heptane) followed by fractional distillation. The chemical composition of each fraction can be measured quantitatively by GC/MS. Fractions can be blended to generate the desired alpha/beta nootkatol and/or nootkatone ingredients for use in flavour or other applications. Verification of acceptability can be carried out by direct comparison to a reference nootkatone flavouring product (for example, an existing natural flavouring commercial product obtained from Frutarom) with analysis provided in Table 9. Two exemplary methods of verification are: 1) Duo-Trio Test, 2) Forced-Choice Preference Test. In one method, the SrKO derived product can be compared to the reference product (for example a commercial Frutarom sourced nootkatone ingredient) in a duo-trio test to determine if the ingredients can be distinguished with statistical significance. This test will determine if the two nootkatone containing ingredients at least match one another based on perception of overall taste and aroma profiles. In the second test, assuming the two products are determined to be distinguishable in a duo trio test, one could determine if the SrKO derived nootkatone is preferred by conducting a forced-choice preference test. More details on these tests are provided as follows. A Duo-Trio Test can be conducted to determine if the blended fractions obtained from the SrKO derived nootkatone flavouring can be distinguished with statistical significance from a reference nootkatone product (for example, a commercially available nootkatone flavouring sourced from Frutarom). The test will determine if the nootkatone flavouring ingredients at least match in terms of overall taste and aroma profile typically conducted in a sugar/acid solution but could also be evaluated in water or sugar water. Methodology: One ounce of the reference sample, labelled “REF” is presented first followed by a one ounce sample of the reference and a one ounce sample of the test sample presented blindly in random order to a minimum of 15 discriminator panellists. The panellists are asked which blind sample is the same as the reference sample. The data are subjected to a statistical analysis to determine the degree of difference between the test sample and the reference control. Assuming a difference is observed between nootkatone flavouring derived from SrKO oxidation and the reference nootkatone product (for example, a Frutarom nootkatone flavouring), a Forced-Choice Preference Test can be conducted to determine if one sample is preferred over the other as a nootkatone flavouring ingredient. The test can be conducted in sugar/acid solution, sugar water or water. Methodology: One ounce of each test sample is presented blindly in random order to a minimum of 40 discriminator panellists. The panellists are asked which blind sample is preferred based on aroma and taste when consumed orally and are forced to make a decision. The data are subjected to a statistical analysis to determine the degree of preference for one sample over the other. To optimize membrane interaction of the initial SrKO variants (referred to in these examples as Valencene Oxidase 1, or VO1), As shown in Mutational analysis of VO1 was conducted in an effort to increase oxygenated titers, as well as to produce altered product profiles. Strain MB2509 (MP6-MEP MP1-ScFPPS Fab46-VS2 MP6-ScCPR) was used as the background, which when transformed with a p5-T7-yhcB-VO1 plasmid produces about 18% nootkatone, about 35% α-nootkatol, and about 47% β-nootkatol, with a complete conversion of valencene. Strains were evaluated for higher production of nootkatone and α-nootkatol. Guided by the homology model based on P450 17A1 (Example 3) site-saturation mutagenesis of the VO active site was conducted at 18 positions, and 5 paired position libraries were constructed. First shell residues were identified through substrate docking, and non-conserved first shell residues were selected based on relative proximity and position for altering the binding pocket geometry. Paired position libraries were constructed by overlap extension PCR and Gibson assembly. Strains were evaluated as in Example 4 for total oxygenation of valencene, and ratio of α- to β-nootkatol. Strains were evaluated at 30° C. and 22° C. Primary screening of paired position libraries revealed that many of the variants lost activity. Library 3 contained variants with improved activity at 22° C. but not 30° C. Thus, introducing two or more mutations simultaneously in the first shell residues can be determimental to activity. Several variants improved oxygenated titers up to 1.7-fold or improved α/β-LGN ratios up to 3.8-fold. Mutations at positions E323, 1390, and Q500 showed several hits with improved oxygenation titer and/or improved α/β profile, and these positions were selected for secondary screening. Next, back-to-consensusmutations (19 mutants) were screened in the VO1 background. Using the screening process described in Example 3, the following mutations were screened: A2T, I389L, I389V, I389A, M94V, T488D, E491K, E52A, H46R, D191N, L150M, I495V, T468I, K344D, Q268T, R351Q, R76K, V400Q, and I444A (numbered according to SEQ ID NO: 37). As shown in Lead variants from active site SSM (L231M, I390L, I390M, T131K, and T131Q), the N-terminal anchor variant n20yhcB_t29VO1, and back-to-consensus mutagenesis were selected, and re-screened. The results of this secondary screen are shown in The seven mutations selected after secondary screening (Example 6) were randomly incorporated into a VO recombination library by allowing either the variant or wild type at each site. The background strain was MB2509 (EGV G2 MP6-CPR)+pBAC-T7-BCD7-yhcB-VO. Primary screening at 30° C. (using the same process described in Example 5) identified several variants with up to 1.35-fold improvement in oxygenated product titers, compared to VOL. Further, select variants showed a shift in production to nootkatone, suggesting higher P450 activity (since production of nootkatone requires two oxygenation cycles). Results of primary screening are shown in The recombination variants were then screened at 34° C. and 37° C. to select leads with improved activity and stability at higher temperature. The results of the secondary screen are shown in A set of cytochrome P450 reductases were screened for improved activity with VO1. This example was done using the strain MB2459 as the background, with pBAC-T7-BCD7-VO1(I382L)-T7BCDx-CPRx. BCD stands for BiCistronic Design, and is described in Mutalik et. al. As shown in The CPR orthologs were retested at 34° C. The results are shown in The ability of alcohol dehydrogenases to convert nootkatols to nootkatone was evaluated. The following ADH enzymes were evaluated: Strains were evaluated as in Example 5, using MB2490 as the background strain (MP6-MEP FAB46-ScFPPS-L-VS1 MP6-VO1-o-SrCPR+p5-T7-BCD14-ADH). Briefly, MP6, Fab46 and T7 refer to the promoter for the attached gene or operon. Here MEP is an operon overexpressing Four orthologs were identified (vvDH, csABA2, bdDH, and zzSDR) that convert (3-nootkatol to nootkatone, resulting in more than a 3-fold increase in nootkatone titers. Protein engineering of cytochrome p450(cam) (CYP101) for the oxidation of polycyclic aromatic hydrocarbons. Protein Eng.. 2000 February; 13(2):121-8.
Chem.. 2005 [cited 2013 May 16]; 3:57-64.
The present disclosure relates to methods for producing oxygenated terpenoids, and preparation of compositions and formulations thereof. Polynucleotides, derivative enzymes, and host cells for use in such methods are also provided. 1. A product comprising:
an oxygenated terpene formulation comprising:
nootkatone in an amount ranging from about 55% to about 60% (w/w); α-nootkatol in an amount ranging from about 20% to about 25% (w/w); and β-nootkatol in an amount ranging from about 5% to about 10% (w/w), wherein the product is a flavor product, a natural flavor product, a fragrance product, a cosmetic product, a cleaning product, a detergent product, a soap product, or a pest control product. 2. The product of 3. The product of 4. The product of 5. The product of 6. The product of 7. A method of making a product for providing oxygenated terpenes, comprising:
obtaining a formulation, said formulation comprising:
nootkatone in an amount ranging from about 55% to about 60% (w/w); α-nootkatol in an amount ranging from about 20% to about 25% (w/w); and β-nootkatol in an amount ranging from about 5% to about 10% (w/w), and incorporating the formulation into a consumer product, an industrial product, a flavor product, a fragrance product, a cosmetic product, a cleaning product, a detergent product, a soap product, or a pest control product. 8. The method of 9. The method of 10. The method of 11. The method of 12. The method of 13. The method of 14. The method of 15. The method of 16. The method of CROSS-REFERENCE TO RELATED APPLICATIONS
FIELD OF THE INVENTION
BACKGROUND OF THE INVENTION
SUMMARY OF THE INVENTION
DESCRIPTION OF THE DRAWINGS
DETAILED DESCRIPTION OF THE INVENTION
# Species Name Native Substrate Native Reaction Product 1 Zingiber zzHO α-humulene 8-hydroxy-α-humulene zerumbet 2 Barnadesia BsGAO germacrene A germacra-1(10),4,11(13)- spinosa trien-12-ol 3 Hyoscyamus HmPO premnaspirodiene solavetivol muticus 4 Latuca LsGAO germacrene A germacra-1(10),4,11(13)- spicata trien-12-ol 5 Nicotiana NtEAO 5-epi-aristolochene capsidiol tabacum 6 Citrus x CpVO valencene nootkatol paradisi 7 Artemesia AaAO amorphadiene artemisinic acid annua 8 Arabidopsis AtKO kaurene kaurenoic acid thaliana 9 Stevia SrKO kaurene kaurenoic acid rebaudiana 10 Pseudomonas PpKO kaurene kaurenoic acid putida 11 Bacillus BmVO fatty acids hydroxylated FAs megateriurn 12 Pleurotus PsVO valencene nootkatone sapidus 13 Pleurotus PoLO unknown unknown ostreatus 14 Cichorium CiVO valencene nootkatone intybus 15 Helianthus HaGAO germacrene A germacrene A acid annuus Summary of some numbered according to wild type S ID NOS: 38 and 106), n22yhcB-t30VO1 (SEQ ID NO: 104), and n22yhcB-t30VO2 (SEQ ID NOS: 61 and 105). Mutation (relative to SEQ Position Position ID NO: 37 (SEQ SEQ ID NO: 37 (SEQ SEQ ID NO: 104 / ID NO: 108) / ID NO: 108) / SEQ ID NO: SEQ ID NO: 38 SEQ ID NO: 38 (SEQ 105 (SEQ ID NO: (SEQ ID NO: No. WT ID NO: 106) 61) 106) 1 L 59 / 47 51 I 2 Y 71 / 59 63 H 3 M 72 / 60 64 K 4 T 75 / 63 67 A 5 A 79 / 67 71 E 6 K 88 / 76 80 R 7 T 92 / 80 84 C 8 M 94 / 82 86 V 9 V 97 / 85 89 L 10 V 97 / 85 89 I 11 S 98 / 86 90 N 12 Q 112 / 100 104 S 13 N 118 / 106 110 K 14 K 124 / 112 116 T 15 A 128 / 116 120 R 16 T 131 / 119 123 S 17 M 135 / 123 127 T 18 M 135 / 123 127 Q 19 M 135 / 123 127 F 20 M 135 / 123 127 T 21 D 139 / 127 131 G 22 Y 141 / 129 133 F 23 A 152 / 140 144 R 24 K 161 / 149 153 R 25 H 162 / 150 154 F 26 N 183 / 171 175 D 27 L 192 / 180 184 F 28 I 195 / 183 187 V 29 D 220 / 208 212 E 30 D 244 / 232 236 E 31 S 279 / 267 271 A 32 H 284 / 272 276 Q 33 S 296 / 284 288 C 34 I 298 / 286 290 L 35 Q 306 / 294 298 K 36 Q 311 / 299 303 E 37 I 322 / 310 314 T 38 I 322 / 310 314 V 39 R 383 / 371 375 K 40 R 383 / 371 375 I 41 V 387 / 375 379 T 42 V 387 / 375 379 I 43 V 387 / 375 379 L 44 I 390 / 378 382 V 45 H 394 / 382 386 Y 46 V 400 / 388 392 Q 47 V 400 / 388 392 M 48 H 405 / 393 397 D 49 L 412 / 400 404 I 50 V 425 / 413 417 D 51 V 425 / 413 417 K 52 F 446 / 434 438 L 53 G 454 / 442 446 A 54 S 462 / 450 454 A 55 L 466 / 454 458 M 56 G 472 / 460 464 A 57 M 476 / 464 468 L 58 M 487 / 475 479 G 59 T 499 / 487 491 N 60 P 504 / 492 496 K 61 I 509 / 497 501 L T 499 / 487 491 S 62 M 135 / 123 127 Q T 499 / 487 491 V 63 M 135 / 123 127 F T 499 / 487 491 V 64 M 135 / 123 127 F T 499 / 487 491 F 65 M 135 / 123 127 F T 499 / 487 491 M 66 M 135 / 123 127 F T 499 / 487 491 G The following mutants were evaluated in the VO1 background (n22-yhcB-t30-VO1, SEQ ID NO: 110) according to wild type S n22yhcB-T30VO1 (SEQ ID NO: 104) and n22yhcB- t30VO2 (SEQ ID NOS: 61 and 105). Mutation (relative to SEQ Position Position ID NO: 37 (SEQ SEQ ID NO: 37 (SEQ SEQ ID NO: 104 / ID NO: 108) / ID NO: 108) / SEQ ID NO: SEQ ID NO: 38 SEQ ID NO: 38(SEQ 105 (SEQ ID (SEQ ID NO: No. WT ID NO: 106) NO: 61) 106) 1 A 2 T 2 H 46/34 38 R 3 E 52/40 44 A 4 R 76/64 68 K 5 M 94/82 86 V 6 T 131/119 123 K 7 T 131/119 123 Q 8 L 150/138 142 M 9 D 191/179 183 N 10 L 231/219 223 M 11 Q 268/256 260 T 12 E 323/311 315 L 13 K 344/332 336 D 14 R 351/339 343 Q 15 I 389/377 381 L 16 I 389/377 381 V 17 I 389/377 381 A 18 I 390/378 382 L 19 I 390/378 382 M 20 V 400/388 392 Q 21 I 444/432 436 A 22 T 468/456 460 I 23 T 488/476 480 D 24 E 491/479 483 K 25 I 495/483 487 V Summary of mutations of several engineered S alignments relative to wild type S each of the S based on the shift value for each sequence relative to wild type S NOS: 38 and 106) is the S and the addition of an 8-amino acid membrane anchor. 8rp-t20VO0 (SEQ ID NO: 107) has a truncation of 20 amino acids of the S terminal anchor, and a single mutation at position 487. n22yhcB-t30VO1 (SEQ ID NO: 104) has a 30-amino acid truncation of the S acids from 480, and 491, and a n22t30-yhcB mutation. n22yhcB-t30VO2 (SEQ ID NOS: 61 and 105) has a 30-amino acid truncation of the S acids from 460, and 491, and a n22t30-yhcB mutation. Wild Type 8rp-t20SrKO n22yhcB- S (SEQ ID 8rp-t20VO0 n22yhcB- t30VO2 (SEQ ID NOS: 37 NOS: 38 and (SEQ ID NO: t30VO1 (SEQ ID NOS: 61 and 108) 106) 107) ID NO: 104) and 105) (Shift Value: (Shift Value: (Shift Value: - (Shift Value: - (Shift Value: - 0) -12) 0) 8) 8) H46 H34 H38R R76 R64 R68K M94 M82 M86V T131 T119 T123Q F231 F219 F223L F223L H284 H272 H276Q H276Q R383 R371 R375K R375K 1390 1378 I382L V400 V388 V392Q 1444 1432 I436A T468 T456 T460I T488 T476 T480D T499 T487 T487N T491N T491N Summary of with respect to wild type (SEQ ID NO: 1) No. WT Position Mutation 1 N 23 D 2 T 37 R 3 P 38 S 4 V 42 R 5 A 45 E 6 C 46 K 7 Q 50 R 8 K 56 E 9 K 59 R 10 R 60 K 11 K 61 M 12 T 63 R 13 T 63 K 14 N 69 Q 15 N 69 K 16 S 71 I 17 Q 72 R 18 L 73 K 19 N 75 E 20 F 76 M 21 F 76 L 22 V 80 M 23 V 80 L 24 V 85 I 25 A 86 S 26 Q 91 D 27 A 96 I 28 Q 98 E 29 Q 98 D 30 C 101 Y 31 N 102 H 32 S 103 D 33 F 104 D 34 F 104 N 35 D 111 E 36 N 116 T 37 I 117 S 38 I 117 V 39 I 117 T 40 G 120 R 41 Q 127 H 42 T 130 N 43 I 131 V 44 I 135 V 45 T 140 K 46 E 142 K 47 E 148 D 48 A 149 S 49 A 149 D 50 I 151 S 51 R 155 K 52 M 157 L 53 G 159 S 54 G 159 N 55 E 162 Q 56 E 162 K 57 A 164 S 58 V 168 T 59 G 170 D 60 L 174 M 61 A 175 E 62 A 175 D 63 K 176 E 64 T 183 K 65 A 187 S 66 M 188 L 67 E 190 N 68 G 193 K 69 A 201 S 70 N 205 E 71 R 206 Q 72 I 208 L 73 R 209 H 74 G 211 R 75 L 212 M 76 E 213 P 77 I 221 L 78 V 223 R 79 Q 225 D 80 Q 225 E 81 D 226 K 82 D 226 E 83 A 228 E 84 F 229 I 85 H 230 V 86 D 231 N 87 K 232 E 88 T 233 A 89 T 233 V 90 S 247 D 91 L 248 M 92 L 248 K 93 K 250 Q 94 E 251 K 95 S 254 K 96 N 255 E 97 A 257 S 98 K 261 A 99 E 262 D 100 D 264 G 101 Y 280 F 102 M 283 I 103 H 284 M 104 H 284 A 105 G 285 A 106 Y 287 F 107 Q 291 N 108 R 294 L 109 R 297 I 110 L 299 M 111 M 305 A 112 M 305 L 113 I 308 M 114 T 318 S 115 P 319 L 116 P 319 I 117 K 323 Q 118 R 331 K 119 D 333 E 120 I 334 E 121 I 334 V 122 N 335 K 123 N 335 Q 124 N 335 S 125 S 336 A 126 Y 343 W 127 Y 348 F 128 V 349 L 129 L 352 I 130 D 353 E 131 D 353 N 132 V 354 T 133 Y 355 F 134 K 356 E 135 K 356 N 136 I 358 V 137 E 359 D 138 E 360 Y 139 E 363 K 140 E 363 L 141 G 366 A 142 Y 369 N 143 R 370 V 144 V 371 I 145 H 372 E 146 H 372 P 147 A 374 G 148 A 374 L 149 E 376 D 150 M 378 I 151 N 380 I 152 N 380 K 153 R 383 Q 154 E 394 Q 155 E 394 D 156 E 395 N 157 E 395 G 158 H 396 Y 159 H 396 Q 160 E 402 D 161 R 405 E 162 C 414 R 163 L 415 M 164 A 417 L 165 T 418 V 166 T 419 H 167 V 422 L 168 M 424 V 169 A 428 V 170 T 429 S 171 T 437 F 172 S 438 G 173 D 439 Y 174 K 441 R 175 I 442 M 176 I 442 L 177 M 443 V 178 S 444 R 179 N 447 S 180 F 448 T 181 M 453 A 182 G 466 E 183 T 469 A 184 Q 478 E 185 Y 479 F 186 G 480 A 187 V 481 A 188 S 482 T 189 Y 487 C 190 S 488 E 191 E 489 H 192 F 490 I 193 F 490 L 194 Q 491 K 195 Q 491 N 196 Q 493 L 197 I 494 M 198 N 496 D 199 D 500 E 200 L 506 M 201 T 509 S 202 V 511 M 203 S 512 P 204 S 512 T 205 M 513 K 206 P 514 D 207 L 519 A 208 D 527 E 209 V 528 F 210 E 532 D 211 Q 533 E 212 Q 533 G 213 S 535 G 214 V 539 S 215 V 542 L 216 V 542 T 217 M 543 I 218 N 546 H 219 V 550 L 220 F 551 L 221 I 552 V 222 N 553 D 223 N 553 E 224 A 554 P 225 V 555 I Summary of mutations evaluated in the Vv2M5 background (SEQ ID NO: 9). No. WT Position Mutation 226 N 18 V 227 V 21 S 228 N 23 D 229 N 27 S 230 Q 32 H 231 I 34 L 232 T 35 S 233 T 37 S 234 K 41 S 235 V 42 E 236 A 45 E 237 K 46 C 238 K 47 M 239 Q 50 R 240 I 51 V 241 D 53 E 242 K 56 E 243 V 67 A 244 A 68 N 245 N 69 D 246 N 69 Q 247 S 71 L 248 Q 72 R 249 V 80 I 250 A 86 S 251 Q 91 K 252 C 101 Y 253 N 102 D 254 N 102 H 255 M 110 D 256 D 111 E 257 G 112 D 258 I 117 S 259 T 130 N 260 R 143 E 261 R 145 N 262 A 149 S 263 S 152 N 264 G 159 N 265 G 159 S 266 V 168 T 267 K 176 E 268 K 186 E 269 A 187 S 270 S 191 H 271 Y 194 P 272 H 195 P 273 N 205 E 274 L 212 M 275 E 213 P 276 W 219 H 277 V 223 I 278 D 226 E 279 A 228 E 280 F 229 S 281 T 233 V 282 V 245 L 283 L 248 M 284 L 256 I 285 K 261 A 286 E 262 D 287 C 347 F 288 E 363 A 289 H 372 E 290 V 377 A 291 E 395 G 292 E 395 N 293 H 396 Y 294 A 399 T 295 C 414 R 296 E 426 D 297 S 438 G 298 M 443 I 299 T 469 A 300 S 488 E 301 K 491 R 302 M 513 K 303 A 517 E 304 L 519 V 305 E 532 D 306 V 550 L 307 N 553 D Summary of mutations evaluated in VS2 background (SEQ ID NO: 11). No. WT Position Mutation 308 P 20 R 309 N 23 D 310 I 28 F 311 K 41 P 312 K 41 S 313 V 42 D 314 R 44 H 315 Q 50 D 316 Q 50 R 317 E 52 R 318 K 61 M 319 N 69 Q 320 Q 72 R 321 L 73 K 322 A 79 I 323 A 86 S 324 H 88 L 325 Q 91 H 326 A 96 I 327 Q 98 R 328 C 101 Y 329 N 102 H 330 C 107 F 331 I 117 S 332 G 120 L 333 T 140 K 334 R 145 N 335 S 152 V 336 V 154 I 337 V 154 P 338 R 155 K 339 M 157 L 340 G 159 M 341 A 175 D 342 K 176 E 343 A 177 P 344 L 178 I 345 H 184 Y 346 H 184 Q 347 A 187 S 348 S 191 H 349 H 195 N 350 L 196 P 351 A 201 R 352 L 212 M 353 E 213 P 354 A 217 Q 355 A 228 E 356 D 231 N 357 K 232 P 358 T 233 V 359 E 236 D 360 D 241 E 361 N 255 D 362 A 257 M 363 L 276 P 364 Y 280 F 365 M 283 I 366 V 286 A 367 T 300 M 368 T 300 I 369 T 306 L 370 T 306 I 371 L 309 I 372 A 315 V 373 E 320 D 374 K 323 R 375 S 336 T 376 E 342 D 377 C 347 L 378 A 350 I 379 N 356 H 380 E 363 G 381 Q 368 P 382 N 380 D 383 Q 381 L 384 E 395 G 385 A 407 G 386 A 407 S 387 C 414 P 388 A 417 I 389 A 432 I 390 V 436 L 391 I 442 P 392 I 442 L 393 S 445 R 394 S 446 M 395 T 450 C 396 S 458 T 397 H 459 Y 398 H 459 M 399 H 467 Q 400 T 469 A 401 E 484 P 402 Q 485 H 403 Q 485 E 404 V 486 A 405 Y 487 L 406 S 488 E 407 I 494 V 408 N 496 D 409 N 496 K 410 M 513 T 411 T 523 I 412 D 527 L 413 I 529 L 414 I 529 M 415 E 532 H 416 E 532 Y 417 S 535 A 418 R 544 H 419 N 546 Y 420 N 546 F 421 A 548 I 422 V 550 L 423 V 555 I EXAMPLES
Example 1: Construction of Sesquiterpene Precursor (Valencene) Producing
Example 2: Functional Activity of CYP450 Library on Valencene Scaffold
Major Products Formed From Valencene by Select P450 Enzymes in SPECIES NAME MAJOR PRODUCTS CiVO β-nootkatol, α-cadinol, hydroxyl valencene. HmPO β-nootkatol, a-cadinol, hydroxyl valencene, nootkatone LsGAO β-nootkatol, α-cadinol, isovalencenol, nootkat-11-en-10-ol. BsGAO β-nootkatol, α-cadinol, isovalencenol. NtEAO α-cadinol, nootkat-11-en-10-ol. SrKO α-nootkatol, hydroxygermacra-1(10)5- diene, β-nootkatol, nootkatone, murolan-3,9(11) diene-10-peroxy ZzHO α-cadinol, nootkat-11-en-10-ol. CpVO α-cadinol, nootkat-11-en-10-ol. MsL6OH α-cadinol. NtVO α-cadinol, nootkat-11-en-10-ol. StVO α-cadinol, β-nootkatol, globulol. AtKO α-cadinol, nootkat-11-en-10-ol. Ci2VO β-nootkatol, α-cadinol, isovalencenol. AaAO α-cadinol, murolol, nootkat-11-en-10- ol. Taxus 5-alpha α-cadinol hydroxylase P450 Example 3: Structural and Mutational Studies of SrKO
BLAST search with SrKO in preparation of Homology Model Sequence Enzyme Name Species Identity Accession kaurene oxidase 99% AAQ63464.1 ent-kaurene oxidase 2 79% BAG71198.1 ent-kaurene oxidase 1 71% BAG71197.1 ent-kaurene oxidase 63% XP_002510288.1 Binding pocket mutations and their fold productivity of total oxygenated oil according to wild type SrKO (SEQ ID NOS: 37 and 108), 8rp-t20SrKO (SEQ ID NOS: 38 and 106), n22yhcB-t30VO1 (SEQ ID NO: 104) and n22yhcB-t30VO2 (SEQ ID NOS: 61 and 105). Mutants Mutants (numbered (numbered according to Mutants according to SEQ ID NO: 104/ (numbered SEQ ID NO: 37) SEQ ID NO: 105 according to (SEQ ID NO: 108) (SEQ ID NO: 61)) Fold Mutant # SEQ ID NO: 38) (Shift value (Shift value productivity (Table (SEQ ID NO: relative to SEQ relative to SEQ (as measured 2.1) 106) ID NO: 38: +12) ID NO.: 38: +4) in mg/L) 38 I310V I322V I314V 1.5 37 I310T I322T I3I4T 0.0 42 V375I V387I V379I 1.4 41 V375T V387T V379T 0.0 19 M123F M135F M127F 0.0 20 M123T M135T M127T 0.3 18 M123Q M135Q M127Q 0.0 59 T487N T499N T491N 2.5 66 M123F_T487G M135F_T499G M127F_T491G 0.0 63 M123F_T487V M135F_T499V M127F_T491V 0.0 62 M123Q_T487V M135Q_T499V M127Q_T491V 0.0 59 T487N_V375F T499N_V387F T491N_V379F 2.2 59 T487N_V375A T499N_V387A T491N_V379F 1.8 59 T487N_V121A T499N_V133A T491N_V125A 2.0 59 T487N_V375M T499N_V387M T491N_V379M 1.9 59 T487N_M120L T499N_M132L T491N_M124L 1.8 59 T487N_M120I T499N_M132I T491N_M124I 1.8 59 T487N_L114V T499N_L126V T491N_L118V 1.4 59 T487N_F219L T499N_F231L T491N_F223L 3.5 59 T487N_M120V T499N_M132V T491N_M124V 1.1 59 T487N_F219I T499N_F231I T491N_F223I 3.3 59 T487N_L114F T499N_L126F T491N_L118F 1.2 Non-binding pocket point mutations and productivity of total oxygenated oil compared to the wild type SrKO (SEQ ID NOS: 38 and 106). Mutants Mutants (numbered (numbered according to according to SEQ ID NO: 37) SEQ ID NO: 104/ Mutants (SEQ ID NO: 108) SEQ ID NO: 105 (numbered (Shift value (SEQ ID NO: 61)) Fold Mutant # according to relative to (Shift value productivity (Table SEQ ID NOS: SEQ ID NO: relative to SEQ (as measured 2.1) 38 and 106) 38: +12) ID NO: 38: +4) in mg/L) 53 G442A G454A G446A 0.849153 55 L454M L466M L458M 0.717318 44 I378V I390V I382V 0.349005 47 V388M V400M V392M 0.792428 9 V85I V97I V89I 0.795913 51 V413K V425K V417K 0.902039 60 P492K P504K P496K 0.131657 40 R371I R383I R375I 0.657808 7 T80C T92C T84C 0.342501 23 A140R A152R A144R 0.014872 2 Y59H Y71H Y63H 0.406787 5 A67E A78E A71E 0.937429 8 M82V M94V M86V 1.585588 11 S86N S98N S90N 0.977752 22 Y129F Y141F Y133F 0.686276 24 K149R K161R K153R 0.990776 29 D208E D220E D212E 0.853446 31 S267A S279A S271A 0.79152 32 H272Q H284Q H276Q 0.958227 33 S284C S296C S288C 0.652348 39 R371K R383K R375K 1.443497 45 H382Y H394Y H386Y 0.609951 46 V388Q V400Q V392Q 0.924043 49 L400I L412I K4041 0.682775 50 V413D V425D V417D 0.039261 52 F434L F446L F438L 0.793926 57 M464L M476L M468L 0.689696 58 M475G M487G M479G 0.573906 61 I497L I509L I501L 0.679949 15 A116R A128R A120R 0.216353 1 L47I L59I L51I 0.88992 25 H150F H162F H154F 0.666723 Example 4: Isolation and Evaluation of Oxygenated Product
SrKO oxidation of valencene Ret. GC-FID Time Compound Name CAS # Area % 33.762 dodecane 112-40-3 6.70 35.440 glyceryl diacetate I 5.26 38.767 triacetin 102-76-1 4.48 39.518 unknown 10.17 40.176 unknown 7.52 42.012 unknown 2.53 44.437 unknown 20.25 44.816 valencene 4630-07-3 1.97 45.546 nootkatene 5090-61-9 1.09 46.260 unknown 2.14 46.395 unknown 6.77 46.869 unknown 4.01 47.394 germacrene D-4-ol 74841-87-5 1.23 48.273 unknown 1.86 49.659 T-muurolol 19912-62-0 0.69 49.753 an unknown sesquiterpene 0.56 50.336 an unknown sesquiterpene 0.58 51.025 epinootkatol (or alpha nootkatol) 50763-66-1 1.96 51.430 Nootkatol (or beta nootkatol) 50763-67-2 3.54 54.138 nootkatone 4674-50-4 15.87 54.501 6-isopropenyl-4,8a-dimethyl- 76784-84-4 0.84 4a,5,6,7,8,8a-hexahydro-2(1H)- naphthalenone TOTAL 100.00 SrKO oxidation of valencene Ret. GC-FID Time Compound Name CAS # Area % 33.763 dodecane 112-40-3 7.26 35.470 glyceryl diacetate I 7.07 38.773 triacetin 102-76-1 6.19 39.526 unknown 11.56 40.179 unknown 8.10 44.440 unknown 23.95 44.821 valencene 4630-07-3 6.88 45.545 nootkatene 5090-61-9 2.22 46.404 unknown 5.08 46.879 unknown 3.66 47.399 germacrene D-4-ol 74841-87-5 2.89 48.279 unknown 2.27 49.665 T-muurolol 19912-62-0 0.94 50.342 an unknown sesquiterpene 1.71 51.027 epinootkatol 50763-66-1 2.48 51.444 nootkatol 50763-67-2 5.24 54.152 nootkatone 4674-50-4 2.49 TOTAL 100.00 Analysis of commercially available natural flavouring nootkatone from Frutarom Ret. GC-FID Time Compound Name CAS # Area % 42.307 limonene glycol 1946-00-5 0.201 42.792 decanoic acid 334-48-5 0.115 49.405 valencene 4630-07-3 0.039 50.362 delta-cadinene 483-76-1 0.268 52.757 alpha-elemol 639-99-6 2.178 53.11 spathulenol 6750-60-3 0.264 53.423 caryophyllene oxide 1139-30-6 0.394 53.748 viridiflorol 552-02-3 0.061 54.225 unknown sesquiterpenoid 0.113 (MW = 220, tent) 54.853 unknown 2.985 55.386 unknown 2.251 55.97 T-muurolol 19912-62-0 0.399 56.192 bulnesol 22451-73-6 0.722 56.523 7(11), 4b-selinenol; tentative 1.425 56.65 unknown (MW = 232, tent) 0.663 56.937 beta-sinensal 3779-62-2 0.914 57.449 unknown 0.285 57.589 cedrenal; tentative 0.438 58.189 unknown sesquiterpenoid(s) 1.077 58.73 unknown sesquiterpenoids 0.537 (MW = 220, 222, tent) 59.102 beta, gamma-nootkatone 35936-67-5 1.805 59.32 myristic acid 544-63-8 0.058 59.537 1,10-dihydronootkatone 20489-53-6 0.582 59.75 a nootkatone isomer 0.442 60.507 nootkatone isomers (2); 0.812 tentative 60.782 unknowns (2) 0.605 61.034 hexadecanal 629-80-1 0.302 62.93 nootkatone 4674-50-4 74.287 63.057 3,11-eudesmadiene-2-one 86917-81-9 1.909 (5S,7R,10R) 63.14 unknown (MW = 234, tent) 0.18 63.26 unknown (MW = 232, tent) 0.105 64.112 heptadecanal 629-90-3 0.344 64.403 unknown sesquiterpenoid 0.446 65.16 unknown sesquiterpenoid 0.147 65.384 palmitic acid 57-10-3 0.154 65.599 alpha-camphorene 532-87-6 0.249 65.75 unknown(s) 0.054 65.878 dehydro-alpha-vetivenone; 0.115 tentative 66.056 nootkatone, 9-oxo 86925-44-2 0.172 66.371 ethyl palmitate 628-97-7 0.185 66.856 cis-9-octadecenal 2423-10-1 0.239 66.986 unknown sesquiterpenoids 0.114 67.556 octadecanal 638-66-4 0.096 74.551 osthol 484-12-8 0.367 80.543 isomerazin 1088-17-1 0.112 84.671 unknown (MW = 298) 0.07 TOTAL 99.28 Duo-Trio Test:
Spring Water 1000 g Citric Acid 0.1% Sucrose 8% Nootkatone flavouring 2.5 ppm Forced-Choice Preference Test:
Spring Water 1000 g Citric Acid 0.1% Sucrose 8% Nootkatone flavouring 2.5 ppm Example 5: N-Terminal Anchor Engineering
Example 6: Mutational Analysis of VO1
Paired Position Libraries (numbered according to SEQ ID NOS: 37 and 108) Library Pos. 1 Allowed AA Pos. 2 Allowed AA 1 V387 F, L, I, S, P, T, A, M P388 S, T, A 2 M132 F, L, I, V, S, P, T, A V133 F, L, I, S, P, T, A, M 3 L123 F, I, V, S, T, A, P, M L126 F, I, V, S, T, A, P, M 4 V387 F, L, I, S, P, T, A, M I322 F, L, V, S, P, A, M, T 5 I322 F, L, V, S, P, A, M, T V133 F, L, I, S, P, T, A, M The following single position SSM was conducted (numbered according to SEQ ID NOS: 37 and 108) Residue Location I390 Channel L392 Channel V387 1st Shell E323 1st Shell I helix I322 1st Shell I helix T499 1st Shell Q500 1st Shell L231 1st Shell F helix L123 1st Shell B-C loop L126 1st Shell B-C loop V125 1st Shell B-C loop V133 1st Shell F87 on BM3 T131 Channel M135 1st Shell L234 1st Shell F helix P238 1st Shell F-G loop M132 1st Shell B-C loop P388 1st Shell Example 7: SrKO Recombination Library Screening
Sets of mutations in lead variants from recombination library (shown relative to SEQ ID NOS: 37 and 108). N20_t29yhcB R76K M94V T131Q L150M I390L T468I A39H Others c9(6) X X X V146L c12(3) X X X X b4(7) X X X X X c11(8) X X X X X X c6(1) X X X X X b6(2) X X X Example 8: Cytochrome P450 Reductase Screening
Example 9: Alcohol Dehydrogenase Enzymes to Alter Product Profile
CPR enzymes Gene UniProtID Organism reCDH Q9RA05 csDH1 A0A067H4B8 csDH2 A0A067H4S0 csDH3 vvDH F6GX78 voDH1 csABA2 A0A067DRA0 csDH A0A0A0KNF1 bdDH I1GLS4 zzSDR F1SWA0 REFERENCES